 Back
BackBiology Midterm Study Guidance: Key Concepts and Practice
Study Guide - Smart Notes

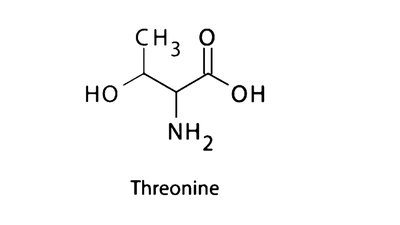
Q9. In the structure of threonine, the asymmetric carbons are attached to the following functional groups EXCEPT:
Background
Topic: Amino Acid Structure and Functional Groups
This question tests your understanding of the structure of amino acids, specifically threonine, and your ability to identify which functional groups are present or absent on its asymmetric (chiral) carbon.
Key Terms and Concepts:
Asymmetric (chiral) carbon: A carbon atom attached to four different groups.
Functional groups: Specific groups of atoms within molecules that have characteristic properties (e.g., methyl, hydroxyl, amino, phosphate).
Step-by-Step Guidance
Examine the structure of threonine and identify the central (chiral) carbon atom.
List all the groups directly attached to this central carbon (look for -CH3, -OH, -NH2, -COOH, etc.).
Compare the list of attached groups to the answer choices: methyl group, hydroxyl group, amino group, phosphate group.
Determine which group is NOT present among those attached to the asymmetric carbon in threonine.
Try solving on your own before revealing the answer!
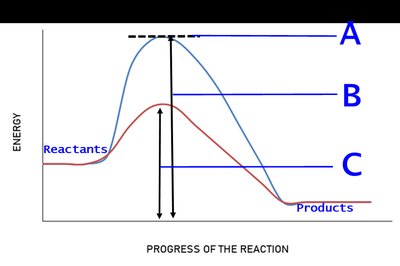
Q16. In the energy diagram for a biological reaction given below, identify the correct labeling for A, B, and C.
Background
Topic: Enzyme Catalysis and Activation Energy
This question tests your ability to interpret an energy diagram for a chemical reaction, specifically how enzymes affect activation energy and the transition state.
Key Terms and Concepts:
Activation energy (Ea): The minimum energy required for a reaction to proceed.
Transition state: The highest energy state during a reaction, where reactants are converted to products.
Enzyme catalysis: Enzymes lower the activation energy, making reactions proceed faster.
Step-by-Step Guidance
Observe the diagram: The y-axis represents energy, and the x-axis represents the progress of the reaction.
Identify the highest point on the curve (A) – this typically represents the transition state.
Compare the two curves: the higher curve (without enzyme) and the lower curve (with enzyme).
Label B and C based on their positions relative to the reactants and the two curves. B is the energy difference for the uncatalyzed reaction, and C is for the catalyzed reaction.
Try solving on your own before revealing the answer!
