 Back
BackCarbon and the Molecular Diversity of Life: Structure and Function of Large Biological Molecules
Study Guide - Smart Notes

Organic Chemistry in Biology
Organic vs Inorganic Compounds
Organic compounds are central to biological systems, distinguished by their carbon backbone and association with living organisms. Inorganic compounds, by contrast, lack this carbon-based structure and are not typically associated with life processes.
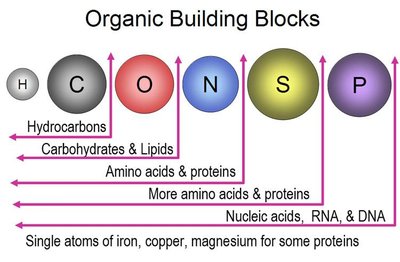
Organic compounds: Contain carbon, often bonded to hydrogen, oxygen, nitrogen, phosphorus, or sulfur.
Inorganic compounds: Include salts, metals, and minerals; do not contain carbon-hydrogen bonds.
Example: Glucose (organic) vs. sodium chloride (inorganic).
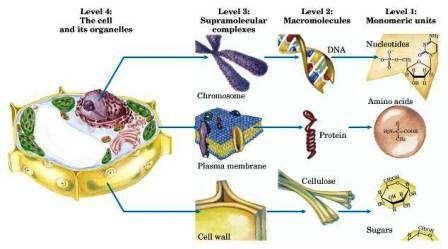
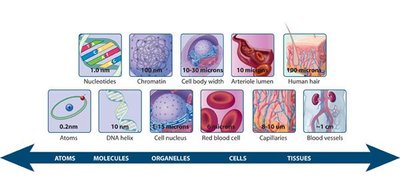
Levels of Biological Organization
Organic molecules serve as the building blocks for higher levels of biological organization, from monomeric units to cells and tissues.
Monomeric units: Sugars, amino acids, nucleotides, fatty acids.
Macromolecules: Polysaccharides, proteins, nucleic acids, lipids.
Supramolecular complexes: Chromosomes, plasma membranes.
Organelles and cells: Cell wall, nucleus, mitochondria.
Structure and Properties of Organic Molecules
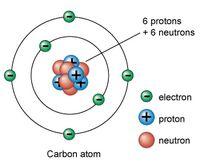
Carbon Atom and Its Bonding
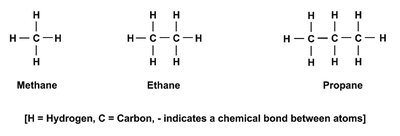
The versatility of carbon arises from its ability to form four covalent bonds, enabling the creation of diverse molecular structures essential for life.
Carbon atom: 6 protons, 6 neutrons, 6 electrons.
Covalent bonding: Allows for stable, complex molecules.
Hydrocarbons: Simplest organic compounds, nonpolar and hydrophobic.


Functional Groups and Isomers
Functional groups are specific clusters of atoms attached to the carbon skeleton, conferring unique chemical properties to organic molecules. Isomers are molecules with the same formula but different structures.
Functional groups: Amino, carbonyl, carboxyl, hydroxyl, phosphate, sulfhydryl.
Properties: Can make molecules polar, acidic, or basic.
Isomers: Structural, geometric, and optical isomers.


Polymers and Monomers
Polymers vs Monomers
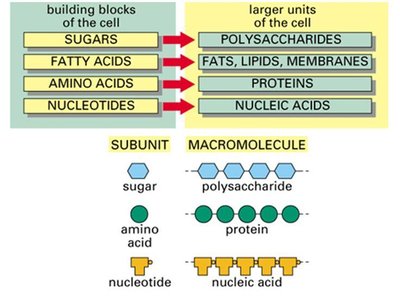
Biological macromolecules are often polymers, composed of repeating monomer units. The assembly and breakdown of these molecules are essential for cellular function.
Monomers: Building blocks (e.g., amino acids, monosaccharides, nucleotides).
Polymers: Large molecules (e.g., proteins, nucleic acids, polysaccharides).






Dehydration and Hydrolysis Reactions
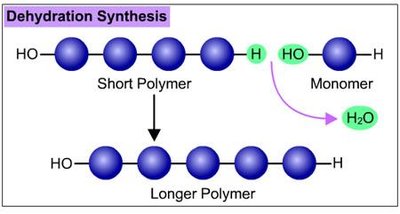
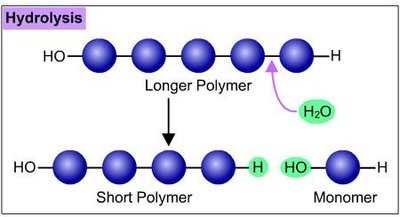
Polymers are synthesized and broken down by dehydration and hydrolysis reactions, respectively. These reactions are fundamental to metabolism and cellular processes.
Dehydration reaction: Joins monomers by removing water; forms polymers.
Hydrolysis reaction: Breaks polymers into monomers by adding water; requires enzymes.






Classes of Organic Molecules
Overview of Four Major Classes
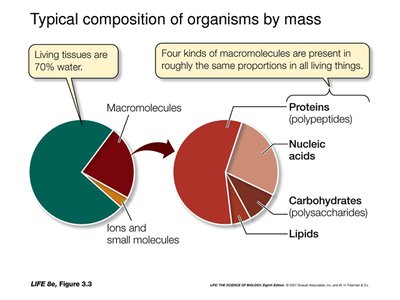
Cells are built from four major classes of organic molecules, each with distinct structures and functions.
Carbohydrates: Energy, energy storage, structural elements.
Lipids: Energy storage, membrane structure, signaling.
Proteins: Catalysis, structure, transport, defense, regulation, motion.
Nucleic acids: Heredity, information storage, protein synthesis.

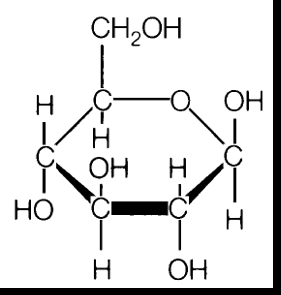
Carbohydrates
Carbohydrates are the most abundant biomolecules, serving as energy sources, energy storage, and structural elements.
Monosaccharides: Simple sugars (e.g., glucose, ribose, fructose).
Disaccharides: Double sugars (e.g., sucrose, lactose).
Polysaccharides: Complex carbohydrates (e.g., starch, glycogen, cellulose, chitin).
Formula:
Lipids
Lipids are hydrophobic molecules, including fats, oils, waxes, phospholipids, and steroids. They serve as energy storage, membrane components, and signaling molecules.
Triglycerides: Glycerol backbone + three fatty acids.
Saturated fatty acids: No double bonds; solid at room temperature.
Unsaturated fatty acids: One or more double bonds; liquid at room temperature.
Phospholipids: Major component of cell membranes; polar head, nonpolar tails.
Steroids: Four fused carbon rings; includes cholesterol, hormones.
Proteins
Proteins are polymers of amino acids, performing diverse functions in cells. Their structure is determined by the sequence and properties of amino acids.
Amino acids: 20 types, each with a unique R group.
Peptide bond: Covalent bond joining amino acids.
Levels of structure: Primary, secondary, tertiary, quaternary.
Denaturation: Loss of structure and function due to environmental changes.
Nucleic Acids
Nucleic acids (DNA and RNA) are polymers of nucleotides, responsible for heredity and protein synthesis.
DNA: Double helix; deoxyribose sugar; bases A, T, G, C.
RNA: Single strand; ribose sugar; bases A, U, G, C.
Central dogma:
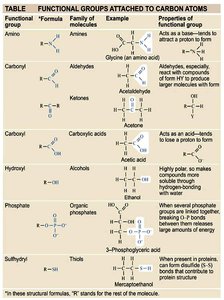
Functional Groups Table
Functional groups attached to carbon atoms determine the chemical reactivity and properties of organic molecules.
Functional Group | Formula | Family of Molecules | Example | Properties |
|---|---|---|---|---|
Amino | -NH2 | Amines | Glycine | Acts as a base |
Carbonyl | -CO | Aldehydes/Ketones | Acetaldehyde/Acetone | Polar, reactive |
Carboxyl | -COOH | Carboxylic acids | Acetic acid | Acidic |
Hydroxyl | -OH | Alcohols | Ethanol | Polar |
Phosphate | -PO4 | Organic phosphates | 3-Phosphoglyceric acid | Energy transfer |
Sulfhydryl | -SH | Thiols | Mercaptoethanol | Forms disulfide bonds |
Summary Table: Building Blocks and Macromolecules
Subunit | Macromolecule |
|---|---|
Sugar | Polysaccharide |
Amino acid | Protein |
Nucleotide | Nucleic acid |

Additional info:
Organic chemistry is foundational to understanding cell structure and function, metabolism, and heredity.
Functional groups are critical for enzyme activity, signaling, and molecular recognition.
Dehydration and hydrolysis reactions are central to metabolic pathways.
