 Back
BackChapter 5: The Working Cell – Membrane Structure, Transport, Energy, and Enzymes
Study Guide - Smart Notes

Membrane Structure and Function
The Fluid Mosaic Model
The plasma membrane is essential for cell survival and function, acting as a boundary and interface for cellular processes. Biologists describe its structure using the fluid mosaic model, which depicts a membrane as a dynamic arrangement of diverse protein molecules suspended in a fluid phospholipid bilayer. This structure allows for selective permeability, enabling the cell to regulate the movement of substances in and out.
Phospholipid bilayer: Provides the fundamental structure, with hydrophobic tails facing inward and hydrophilic heads facing outward.
Membrane proteins: Serve various functions, including transport, signaling, and structural support.
Selective permeability: Only certain molecules can cross the membrane freely, while others require assistance.

Example: Transport proteins embedded in the membrane facilitate the movement of ions and nutrients.
Membrane Formation and Evolution
Spontaneous Formation of Membranes
Phospholipids spontaneously self-assemble into bilayers, forming simple membranes. This process was a critical step in the origin of life, as it allowed for the compartmentalization of molecules and the development of the first cells.
Self-assembly: Driven by the amphipathic nature of phospholipids.
Evolutionary significance: Membrane-enclosed collections of molecules enabled cellular complexity.
Additional info: Membranes also require proteins for transport and signaling, not just lipids.
Transport Across Membranes
Passive Transport: Diffusion
Diffusion is the movement of particles from an area of higher concentration to lower concentration. When diffusion occurs across a cell membrane without energy input, it is called passive transport.
Key Point: Passive transport relies on concentration gradients and does not require cellular energy.
Example: Oxygen and carbon dioxide diffuse freely across cell membranes.
Osmosis: Diffusion of Water
Osmosis is the diffusion of water across a selectively permeable membrane. Water moves from areas of lower solute concentration to higher solute concentration until equilibrium is reached.
Key Point: Osmosis is crucial for maintaining cellular water balance.
Example: In plant cells, osmosis helps maintain turgor pressure.
Tonicity and Water Balance
Tonicity describes the ability of a solution to cause a cell to gain or lose water. Cells respond differently to hypertonic, hypotonic, and isotonic environments:
Hypertonic: Cells lose water and shrink.
Hypotonic: Cells gain water and swell.
Isotonic: No net water movement; animal cells remain normal, plant cells become flaccid.
Facilitated Diffusion
Polar or charged substances cannot easily cross the membrane. Facilitated diffusion uses transport proteins to move these substances down their concentration gradient without energy input.
Transport proteins: Provide channels or carriers for specific molecules.
Aquaporins: Specialized channels for rapid water transport.
Additional info: Facilitated diffusion is essential for glucose and ion transport in cells.
Active Transport
Active transport requires energy (usually from ATP) to move solutes against their concentration gradient. This process is vital for maintaining cellular ion balances.
Key Point: Active transport enables cells to concentrate substances internally or externally.
Example: Sodium-potassium pump maintains electrochemical gradients in animal cells.
Bulk Transport: Exocytosis and Endocytosis
Cells use exocytosis to export large molecules and endocytosis to import them. Endocytosis includes phagocytosis (engulfing particles) and receptor-mediated endocytosis (specific uptake).
Vesicles: Membrane-bound sacs transport material across the membrane.
Example: White blood cells use phagocytosis to engulf pathogens.
Energy and the Cell
Forms of Energy and Thermodynamics
Energy is the capacity to cause change. Cells use kinetic energy (motion), potential energy (stored), and chemical energy (in molecules). The laws of thermodynamics govern energy transformations:
First law: Energy cannot be created or destroyed, only transformed.
Second law: Energy transformations increase disorder (entropy), with some energy lost as heat.
Additional info: Cellular processes rely on energy coupling to drive endergonic reactions.
Chemical Reactions: Exergonic and Endergonic
Exergonic reactions release energy, while endergonic reactions require energy input. Metabolism encompasses all chemical reactions in a cell.
Example: Cellular respiration is exergonic; photosynthesis is endergonic.
ATP and Cellular Work
ATP (adenosine triphosphate) is the cell’s energy currency. It powers cellular work by transferring a phosphate group to other molecules, coupling exergonic and endergonic reactions.
ATP hydrolysis:
ATP cycle: ATP is regenerated from ADP and phosphate using energy from cellular respiration.
Example: Muscle contraction and active transport are powered by ATP.
How Enzymes Function
Enzyme Catalysis
Enzymes are biological catalysts that speed up chemical reactions by lowering the activation energy barrier. They are not consumed in the reaction.
Activation energy: The energy required to start a reaction.
Enzyme specificity: Each enzyme catalyzes a specific reaction.
Example: Amylase catalyzes the breakdown of starch into sugars.
The Catalytic Cycle of Enzymes
An enzyme’s substrate fits into its active site, forming an enzyme-substrate complex. The enzyme facilitates the conversion of substrates to products, then releases the products and is ready for another cycle.
Induced fit: The enzyme changes shape slightly to accommodate the substrate.
Products: The result of the enzymatic reaction.
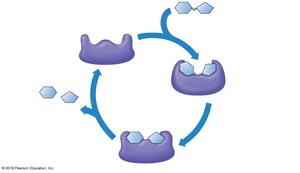
Example: The breakdown of hydrogen peroxide by catalase.
Enzyme Inhibition and Regulation
Enzyme activity can be regulated by inhibitors:
Competitive inhibitors: Bind to the active site, blocking substrate access.
Noncompetitive inhibitors: Bind elsewhere, changing the enzyme’s shape and reducing activity.
Feedback inhibition: The end product of a pathway inhibits an earlier enzyme, regulating metabolism.
Example: Feedback inhibition controls amino acid synthesis in cells.
Enzyme Inhibitors: Drugs, Pesticides, and Poisons
Many drugs, pesticides, and poisons act as enzyme inhibitors. Their effects depend on whether inhibition is reversible or irreversible.
Reversible inhibition: Temporary and can be overcome.
Irreversible inhibition: Permanent, often due to covalent bonding.
Example: Aspirin inhibits cyclooxygenase, reducing inflammation.
Summary Table: Types of Membrane Transport
Type | Energy Required? | Direction | Example |
|---|---|---|---|
Passive Transport | No | Down gradient | Oxygen diffusion |
Facilitated Diffusion | No | Down gradient | Glucose transport |
Active Transport | Yes (ATP) | Against gradient | Sodium-potassium pump |
Osmosis | No | Water down gradient | Plant cell turgor |
Exocytosis | Yes | Out of cell | Protein secretion |
Endocytosis | Yes | Into cell | Phagocytosis |
Additional info: Transport proteins are crucial for selective permeability and cellular homeostasis.
