 Back
BackChapter 8: Reactions in Aqueous Solutions – Chemistry of the Hydrosphere (Biology Context)
Study Guide - Smart Notes

Reactions in Aqueous Solutions: Chemistry of the Hydrosphere
Introduction
This chapter explores the fundamental chemical processes that occur in aqueous solutions, with a focus on their relevance to biological and environmental systems. Understanding these reactions is essential for biology students, as many physiological and ecological processes depend on the chemistry of water-based solutions.
Solutions and Their Concentrations
Key Terminology
Solution: A homogeneous mixture of two or more substances.
Solvent: The component present in the greatest amount; in aqueous solutions, this is water.
Solute: Any component in a solution other than the solvent.
In biological systems, water is the universal solvent, making aqueous solutions central to life processes.
Concentration Units
Mass-to-mass ratios: Used for very dilute solutions, such as parts per million (ppm).
Mole-to-volume ratio (Molarity, M): Molarity (M) is defined as moles of solute per liter of solution.
Common units: mg solute/kg solvent, mg solute/L water, moles solute/L solution.
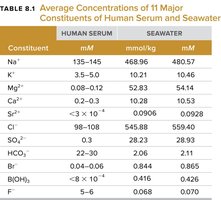
Table 8.1 compares the concentrations of major ions in human serum and seawater, highlighting the biological importance of solution chemistry.
Molarity Calculations
To prepare a solution of known molarity, calculate the required mass of solute using the formula:
Example: To make 10.0 L of 10.0 mM Na2HPO4, calculate moles needed and convert to grams using molar mass.
Dilutions
Principles of Dilution
Dilution involves adding solvent to a solution to decrease its concentration without changing the amount of solute.
The relationship is given by:
Standard solutions are used to prepare solutions of lower concentrations for experiments and clinical use.
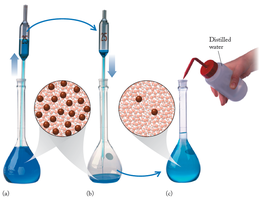
This diagram illustrates the process of dilution, showing how the number of solute particles remains constant while the volume increases.
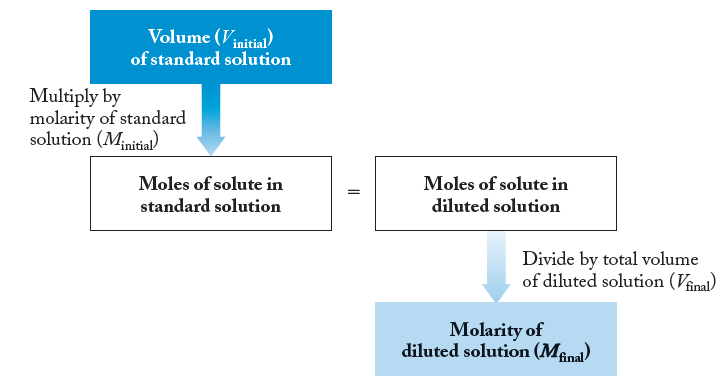
This flowchart summarizes the calculation steps for determining the final molarity after dilution.
Beer’s Law and Concentration Determination
The intensity of a solution’s color is proportional to its concentration, described by Beer’s Law:
A: Absorbance
\varepsilon: Molar absorptivity
b: Path length
c: Concentration
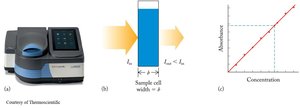
This image shows a spectrophotometer and the linear relationship between absorbance and concentration.
Electrolytes and Nonelectrolytes
Definitions and Examples
Electrolytes: Substances that dissociate into ions in water, conducting electricity.
Strong electrolytes: Completely dissociate (e.g., NaCl, HCl).
Weak electrolytes: Partially dissociate (e.g., acetic acid, ammonia).
Nonelectrolytes: Do not dissociate; do not conduct electricity (e.g., sugar, ethanol).

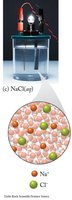

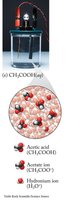

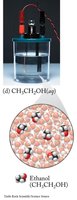
These images compare the molecular and ionic composition of strong electrolytes, weak electrolytes, and nonelectrolytes in solution.
Acid–Base Reactions: Proton Transfer
Brønsted–Lowry Definitions
Acids: Proton (H+) donors
Bases: Proton acceptors
Example:
Neutralization Reactions
When an acid reacts with a base, a salt and water are formed:
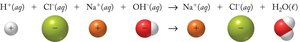
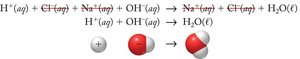
These images show the stepwise breakdown from molecular to net ionic equations, emphasizing the removal of spectator ions.
Strong and Weak Acids/Bases
Strong acids: Completely ionize in water (e.g., HCl, HNO3, H2SO4).
Weak acids: Partially ionize (e.g., acetic acid, HF).
Strong bases: Group 1 and 2 metal hydroxides (e.g., NaOH).
Weak bases: Partially ionize (e.g., NH3).
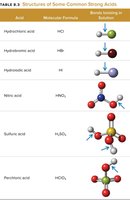
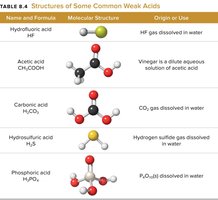
Tables summarize the structures and examples of common strong and weak acids.
Titrations
Principles of Titration
Titration is used to determine the concentration of a solute by reacting it with a standard solution of known concentration. The endpoint is detected by a color change (indicator).


These images show the titration process and the visual endpoint detection.
Precipitation Reactions
Formation of Precipitates
Precipitation reactions occur when soluble reactants form an insoluble product (precipitate). Solubility rules help predict the formation of precipitates.
Example:


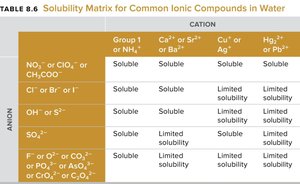
These tables and diagrams summarize solubility rules and the outcomes of mixing various ionic compounds.
Types of Solutions
Saturated: Contains the maximum amount of solute at a given temperature.
Unsaturated: Contains less than the maximum amount of solute.
Supersaturated: Contains more than the maximum predicted amount; unstable.

This image shows the crystallization process in a supersaturated solution.
Oxidation–Reduction (Redox) Reactions: Electron Transfer
Definitions and Principles
Oxidation: Loss of electrons (increase in oxidation number).
Reduction: Gain of electrons (decrease in oxidation number).
Redox reactions involve the transfer of electrons between species.
Assigning Oxidation Numbers
Rules for assigning oxidation numbers help identify which atoms are oxidized or reduced.

This table summarizes the rules for assigning oxidation numbers to atoms in compounds and ions.
Redox Agents
Oxidizing agent: Accepts electrons and is reduced.
Reducing agent: Donates electrons and is oxidized.
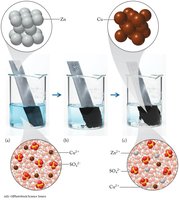
This diagram illustrates the electron transfer between a reducing and oxidizing agent.
Balancing Redox Reactions
Redox reactions are balanced by separating into half-reactions, balancing atoms and charges, and combining the half-reactions.
Example:
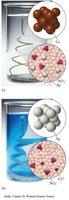
This image shows the stepwise balancing of a redox reaction using the half-reaction method.
Activity Series for Metals
The activity series ranks metals by their ability to act as reducing agents. A metal higher in the series will reduce the ions of metals below it.
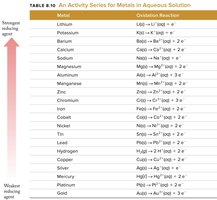
This table lists metals in order of reducing strength, useful for predicting redox reaction outcomes.
Redox Reactions in Acidic and Basic Solutions
Redox reactions can occur in acidic or basic solutions, requiring special balancing steps for H+ or OH− ions.
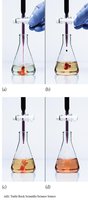
This image shows the endpoint of a redox titration, indicated by a color change.
Summary Table: Solubility Guidelines for Common Ionic Compounds in Water
Rule | Description |
|---|---|
1 | All compounds containing group 1 ions (alkali metals) and NH4+ are soluble. |
2 | Compounds with NO3−, ClO4−, or CH3COO− are soluble. |
3 | Most Cl−, Br−, and I− salts are soluble except with Ag+, Hg22+, or Pb2+. |
4 | SO42− salts are soluble except with Ca2+, Sr2+, Ba2+, Pb2+, Hg22+, or Ag+. |
5 | CO32−, PO43−, S2−, and OH− compounds are generally insoluble except with group 1 ions and NH4+. |
Additional info: These guidelines are essential for predicting precipitation reactions in biological and environmental systems.
Conclusion
Understanding reactions in aqueous solutions is fundamental for biology students, as these processes underpin physiological functions, environmental chemistry, and laboratory techniques. Mastery of solution concentration, acid-base and redox reactions, and solubility rules is essential for interpreting and predicting chemical behavior in biological contexts.
