 Back
BackLEC 2: Classification of Life, Domains, and the Chemical Basis of Biology
Study Guide - Smart Notes

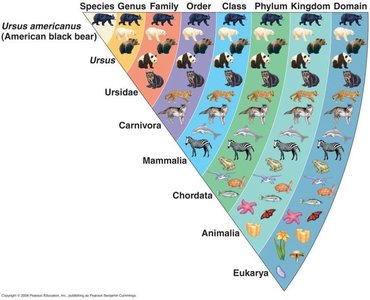
Classification of Living Things: Taxonomy
Hierarchical System of Classification
Taxonomy is the science of classifying living organisms into hierarchical groups based on shared characteristics. The modern system, developed by Carolus Linnaeus, uses a series of ranked categories called taxa. Each organism is given a unique binomial name (Genus species), such as Saccharomyces cerevisiae for brewer’s yeast.
Domain – The broadest category (e.g., Eukarya, Bacteria, Archaea)
Kingdom – Groups of related phyla (e.g., Animalia, Plantae, Fungi, Protista, Bacteria, Archaea)
Phylum – Groups of related classes
Class – Groups of related orders
Order – Groups of related families
Family – Groups of related genera
Genus – Groups of related species
Species – The most specific level; organisms that can interbreed and produce fertile offspring
The Three Domains of Life
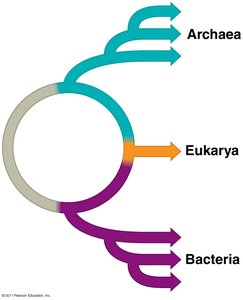
Woese’s Three-Domain System
All living organisms are classified into three domains based on genetic and cellular differences: Bacteria, Archaea, and Eukarya. This system reflects evolutionary relationships and major differences in cell structure.
Bacteria: Prokaryotic, single-celled organisms with peptidoglycan in their cell walls.
Archaea: Prokaryotic, often extremophiles, with unique membrane lipids and no peptidoglycan.
Eukarya: Eukaryotic cells with membrane-bound organelles; includes animals, plants, fungi, and protists.
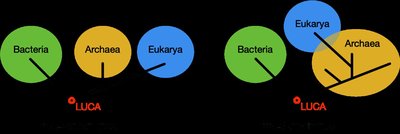
Alternative Models of Early Evolution
Recent genomic data suggest that the evolutionary history of life may be more complex than a simple tree, with significant lateral gene transfer and possible fusion events. The "Ring of Life" and "Two-Domain" models propose that Eukaryotes arose from a fusion of specific Archaea and Bacteria, challenging the traditional three-domain tree.
Summary Table: Key Features of the Three Domains
Feature | Bacteria | Archaea | Eukarya |
|---|---|---|---|
Nuclear Envelope | No | No | Yes |
Membrane-bound Organelles | No | No | Yes |
Peptidoglycan in Cell Wall | Yes | No | No |
Circular Chromosome(s) | Yes | Yes | No (linear) |
Typical Reproduction | Asexual (binary fission) | Asexual (binary fission) | Sexual (mitosis/meiosis) |
The Chemical Context of Life
Elements and Matter
All living organisms are composed of matter, which consists of elements. Of the 92 naturally occurring elements, about 25 are essential for life. The most abundant elements in living matter are carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P), and sulfur (S).
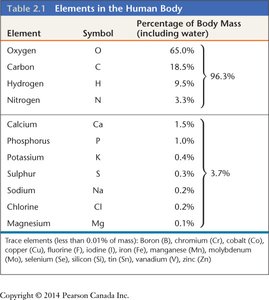
Major Elements in the Human Body
Most of the human body is made up of a few key elements. The table below summarizes their relative abundance:
Element | Symbol | Percentage of Body Mass |
|---|---|---|
Oxygen | O | 65.0% |
Carbon | C | 18.5% |
Hydrogen | H | 9.5% |
Nitrogen | N | 3.3% |
Calcium | Ca | 1.5% |
Phosphorus | P | 1.0% |
Potassium | K | 0.4% |
Sulfur | S | 0.3% |
Sodium | Na | 0.2% |
Chlorine | Cl | 0.2% |
Magnesium | Mg | 0.1% |
Atomic Structure and Chemical Bonds
Atomic Number and Mass
The atomic number of an element is the number of protons in its nucleus, which also equals the number of electrons in a neutral atom. The atomic mass is the sum of protons and neutrons. The arrangement of electrons in discrete shells determines the chemical properties of the atom, especially the valence electrons in the outermost shell.
Electron Shells and Orbitals
Electrons occupy specific energy levels or shells. The first shell holds up to 2 electrons, and the second shell holds up to 8. The distribution of electrons in these shells and their orbitals (s and p) influences how atoms interact and bond.


Covalent Bonds
A covalent bond involves the sharing of a pair of valence electrons between two atoms. Molecules are formed when atoms are held together by covalent bonds. Covalent bonds can be single (one pair shared) or double (two pairs shared). The polarity of covalent bonds depends on the difference in electronegativity between the atoms involved.
Nonpolar covalent bond: Electrons are shared equally (e.g., H2, O2).
Polar covalent bond: Electrons are shared unequally, resulting in partial charges (e.g., H2O).
Ionic Bonds
Ionic bonds form when one atom donates an electron to another, resulting in oppositely charged ions (cations and anions) that attract each other. Ionic compounds, such as salts, dissociate into ions in water.
Weak Chemical Interactions
Hydrogen bonds: Form between a hydrogen atom covalently bonded to an electronegative atom (like O or N) and another electronegative atom. These are crucial in stabilizing the structures of proteins and nucleic acids.
Van der Waals interactions: Weak attractions due to transient local partial charges; important in large molecules and biological surfaces (e.g., gecko feet adhesion).
Properties of Water
Unique Properties of Water
Water is a polar molecule with several properties essential for life:
Cohesion: Water molecules stick together via hydrogen bonds, aiding transport in plants.
Moderation of temperature: Water has a high specific heat and heat of vaporization, helping organisms resist temperature changes and cool via evaporation.
Lower density of ice: Solid water (ice) is less dense than liquid water, so ice floats, insulating aquatic life in winter.
Solvent properties: Water dissolves many polar and ionic substances, making it the "universal solvent" for biological reactions.
Hydrophilic and Hydrophobic Substances
Polar molecules and ions are hydrophilic (water-soluble), while nonpolar molecules are hydrophobic (not water-soluble). This distinction is fundamental to the structure of cell membranes and the behavior of biological molecules in aqueous environments.
Additional info: The notes above cover content from Ch. 1 (Themes of Biology, Scientific Inquiry), Ch. 2 (Chemical Context of Life), and Ch. 3 (Water and Life) of a typical college biology course.
