 Back
BackFeedback Circuits Regulating Digestion, Energy Storage, and Appetite
Study Guide - Smart Notes

Feedback Circuits Regulating Digestion, Energy Storage, and Appetite
Regulation of Digestion
Animals regulate the activity of their digestive systems to match their nutritional needs and energy demands. Digestive processes are activated in a stepwise manner, with each compartment of the digestive tract responding to the presence of food by secreting digestive juices and initiating muscular contractions to move food along.
Nervous Regulation: The enteric nervous system, a network of neurons dedicated to the digestive organs, coordinates reflexes such as saliva secretion and swallowing.
Endocrine Regulation: Hormones released by the stomach and duodenum (e.g., gastrin) ensure that digestive secretions are produced only when needed. These hormones travel through the bloodstream to their target organs.
Example: The arrival of food in the stomach triggers the release of gastric juices and churning, while movement through the intestines is tightly regulated by both neural and hormonal signals.
Regulation of Energy Storage
Animals store excess energy in the form of glycogen (in liver and muscle) and triglycerides (in adipose tissue). Glycogen stores are limited, but fat stores are not, making adipose tissue the most efficient long-term energy reserve.
Energy Storage: Excess energy is stored as glycogen or fat. Glycogen is a polysaccharide composed of glucose units, while triglycerides are stored in adipose cells.
Energy Mobilization: During fasting or heavy exercise, glycogen and fat reserves are mobilized to provide glucose and fatty acids for cellular respiration.
Energy Yield: Fats yield about twice as much energy per gram as carbohydrates or proteins.
Example: Bears accumulate large fat reserves before hibernation to sustain them through periods without food.
Glucose Homeostasis
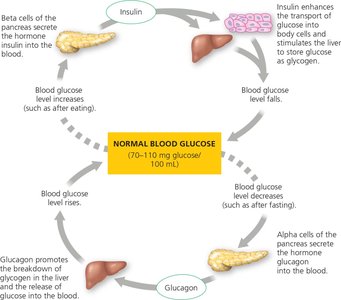
Maintaining blood glucose within a narrow range (70–110 mg/100 mL) is essential for metabolic balance. This is achieved through the antagonistic actions of the hormones insulin and glucagon, both produced by the pancreas.
Insulin: Secreted by beta cells in the pancreas when blood glucose rises (e.g., after eating). Insulin promotes glucose uptake by cells and stimulates the liver to store glucose as glycogen, lowering blood glucose levels.
Glucagon: Secreted by alpha cells in the pancreas when blood glucose falls (e.g., during fasting). Glucagon stimulates the liver to break down glycogen and release glucose into the blood, raising blood glucose levels.
Example: After a meal, insulin ensures excess glucose is stored, while between meals, glucagon ensures glucose is available for cellular respiration.
Pancreatic Hormones and Blood Glucose Regulation
The pancreas contains clusters of endocrine cells called islets, which include alpha cells (glucagon) and beta cells (insulin). These hormones are released into the bloodstream to regulate glucose homeostasis.
Insulin: Stimulates glucose uptake in most body cells (except brain cells) and promotes glycogen synthesis in the liver.
Glucagon: Stimulates glycogen breakdown and gluconeogenesis in the liver, releasing glucose into the blood.
Dual Role of Pancreas: The pancreas functions as both an endocrine (hormone-secreting) and exocrine (digestive enzyme-secreting) organ.
Diabetes Mellitus
Diabetes mellitus is a disorder of glucose homeostasis caused by insufficient insulin production or impaired insulin response. It leads to elevated blood glucose levels and can cause serious complications.
Type 1 Diabetes (Insulin-dependent): An autoimmune disorder where the immune system destroys pancreatic beta cells, eliminating insulin production. Treated with insulin injections.
Type 2 Diabetes (Non-insulin-dependent): Characterized by insulin resistance in target cells. Risk factors include obesity and inactivity. Managed with diet, exercise, and sometimes medication.
Consequences: High blood glucose can damage blood vessels, nerves, kidneys, and eyes. In severe cases, fat metabolism leads to acidosis and electrolyte imbalance.
Example: Type 2 diabetes is a growing public health concern, often associated with obesity and sedentary lifestyles.
Regulation of Appetite and Consumption
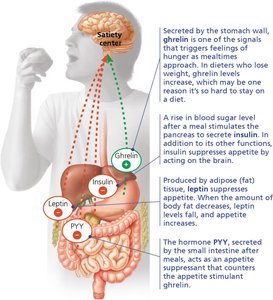
Appetite and energy intake are regulated by a network of hormones and neural circuits that communicate the body's energy status to the brain's satiety center.
Ghrelin: Secreted by the stomach, stimulates hunger before meals.
Insulin and PYY: Both suppress appetite after meals. PYY is secreted by the small intestine.
Leptin: Produced by adipose tissue, suppresses appetite and regulates long-term body fat levels.
Obesity: Chronic overnourishment leads to excessive fat accumulation, increasing the risk of type 2 diabetes, cardiovascular disease, and other health problems.
Example: During the COVID-19 pandemic, obesity was identified as a major risk factor for severe outcomes.
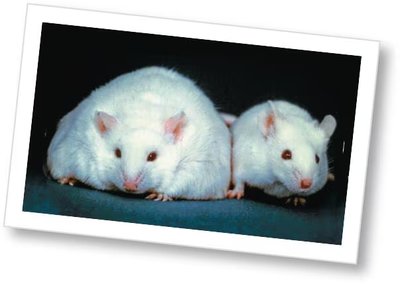
Genetic Regulation of Appetite: The ob and db Genes
Mutations in the ob and db genes in mice disrupt normal appetite regulation, leading to obesity. These genes are involved in the leptin signaling pathway, which suppresses appetite when energy stores are sufficient.
ob Gene: Encodes leptin, a hormone that signals satiety.
db Gene: Encodes the leptin receptor. Mutations in either gene result in excessive eating and obesity.
Experimental Evidence: Parabiosis experiments (surgically joining circulatory systems) show that leptin or its receptor is necessary for appetite suppression.
Experimental Data: Parabiosis and Appetite Regulation
The following table summarizes the results of pairing mice with different genotypes to study the effects of circulating factors on body mass:
Genotype Pairing (red type indicates mutant genes) | Average Change in Body Mass of Subject (g) |
|---|---|
(a) ob+/ob+, db+/db+ paired with ob+/ob+, db+/db+ | 8.3 |
(b) ob/ob, db+/db+ paired with ob+/ob+, db+/db+ | 38.7 |
(c) ob/ob, db+/db+ paired with ob+/ob+, db+/db+ | 8.2 |
(d) ob/ob, db/db paired with ob+/ob+, db+/db/db | −14.9* |
*Due to pronounced weight loss and weakening, subjects in this pairing were remeasured after less than eight weeks.
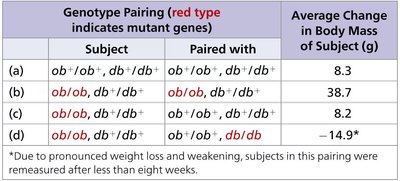
Interpretation: These results demonstrate that the ob gene product (leptin) suppresses appetite, and that the db gene is required for the response to leptin. Mutations in either gene disrupt normal appetite regulation, leading to obesity or, in some cases, excessive weight loss.
Summary Table: Key Hormones in Energy and Appetite Regulation
Hormone | Source | Main Effect |
|---|---|---|
Insulin | Pancreatic beta cells | Lowers blood glucose, suppresses appetite |
Glucagon | Pancreatic alpha cells | Raises blood glucose |
Leptin | Adipose tissue | Suppresses appetite, regulates fat stores |
Ghrelin | Stomach | Stimulates hunger |
PYY | Small intestine | Suppresses appetite after meals |
Key Equations
Glycogen Synthesis:
Glycogen Breakdown:
Additional info: The regulation of appetite and energy balance is a complex interplay between the nervous and endocrine systems, with feedback circuits ensuring homeostasis. Disruptions in these circuits can lead to metabolic diseases such as obesity and diabetes mellitus.
