 Back
BackLEC 20:Genes, Proteins, and the Flow of Genetic Information
Study Guide - Smart Notes

Proteins: Structure and Function
Primary Structure of Proteins
The primary structure of a protein is the unique sequence of amino acids in a polypeptide chain. This sequence determines the protein's final shape and properties, as the order of amino acids dictates how the chain will fold and interact with itself and other molecules.
Peptide bonds link amino acids together, forming a polypeptide.
The sequence is encoded by the gene corresponding to the protein.
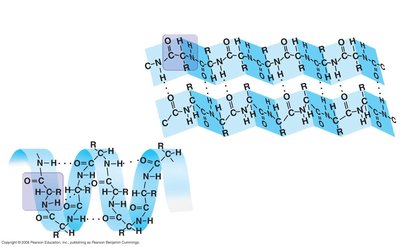
Secondary Structure of Proteins
The secondary structure refers to localized folding or coiling of the polypeptide chain, stabilized by hydrogen bonds between atoms in the backbone (not the R groups). The two main types are the alpha-helix and the beta-pleated sheet.
Alpha-helix (α-helix): A right-handed coil stabilized by hydrogen bonds.
Beta-pleated sheet (β-sheet): Sheet-like structure formed by hydrogen bonds between parallel or antiparallel segments.
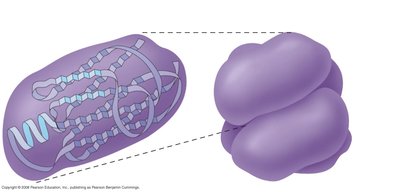
Tertiary Structure of Proteins
The tertiary structure is the overall three-dimensional shape of a single polypeptide chain, resulting from interactions between R groups (side chains) of amino acids. These interactions include hydrophobic interactions, hydrogen bonds, ionic bonds, and covalent disulfide bridges (between cysteine residues).
Determines the protein's functional properties.
Stabilized by various chemical interactions.
Quaternary Structure of Proteins
The quaternary structure exists in proteins composed of more than one polypeptide chain. These chains (subunits) assemble into a functional protein complex. Not all proteins have quaternary structure.
Examples: Hemoglobin (4 subunits), DNA polymerase (multiple subunits).
Protein complexes can include different proteins associating together.

Genes and the Relationship to Proteins
Definition and History of the Gene
A gene is a unit of hereditary information. It can be defined as:
A functional unit of heredity that determines an observable trait (phenotype).
A DNA sequence that encodes the chemical structure of a specific polypeptide or RNA molecule.
Early ideas of heredity suggested blending of parental traits, but this could not explain the persistence of traits over generations. Gregor Mendel proposed that inheritance is particulate, with discrete units (now called genes) passed from parent to offspring.

Mendelian Inheritance
Mendel's experiments with pea plants demonstrated that traits are inherited as discrete units (genes), not by blending. He observed dominant and recessive traits, and formulated the concept of paired factors (alleles).
Dominant traits mask recessive traits in hybrids.
Recessive traits reappear in predictable ratios (e.g., 3:1 in F2 generation).
Genes and Proteins: The One Gene-One Enzyme Hypothesis
Sir Archibald Garrod first suggested that genes are responsible for the production of enzymes, linking genotype to phenotype. This was supported by Beadle and Tatum's experiments with Neurospora crassa, showing that each gene encodes a specific enzyme (later generalized to polypeptides).
Mutations in genes can lead to loss of enzyme function, causing metabolic disorders (e.g., alkaptonuria).
Not all genes encode polypeptides; some encode functional RNAs (e.g., rRNA, tRNA).


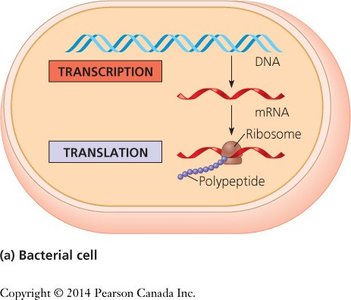
The Central Dogma of Molecular Biology
Overview of Information Flow
The central dogma describes the flow of genetic information in cells: DNA is transcribed into RNA, which is then translated into protein. This process involves two main steps:
Transcription: Synthesis of RNA from a DNA template.
Translation: Synthesis of a polypeptide from an mRNA template, using ribosomes.
In eukaryotes, transcription occurs in the nucleus and translation in the cytoplasm. In prokaryotes, both processes occur in the same compartment.
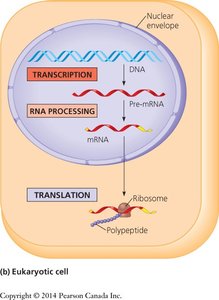
Types of RNA
mRNA (messenger RNA): Encodes the amino acid sequence of a protein.
tRNA (transfer RNA): Brings amino acids to the ribosome during translation.
rRNA (ribosomal RNA): Structural and catalytic component of ribosomes.
The Genetic Code
Codons and the Triplet Code
The genetic code is read in sets of three nucleotides called codons. Each codon specifies a particular amino acid or a stop signal during translation. The code is nearly universal among organisms.
There are 64 possible codons (43 combinations).
Some amino acids are specified by more than one codon (redundancy).
Three codons (UAA, UAG, UGA) are stop codons; AUG is both the start codon and codes for methionine.
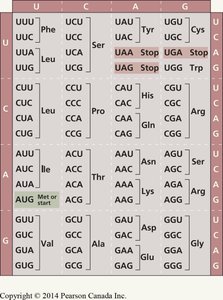
Reading Frame and Translation
The correct reading frame is essential for translating the genetic code accurately. Translation begins at a start codon (AUG) and proceeds codon by codon until a stop codon is reached. The mRNA is read in the 5′→3′ direction, and the resulting polypeptide is synthesized from the amino (N) to the carboxyl (C) terminus.
The number of nucleotides in an mRNA is at least three times the number of amino acids in the protein.
Mutations that shift the reading frame (frameshift mutations) can drastically alter the resulting protein.
Summary Table: Levels of Protein Structure
Level | Description | Stabilizing Interactions |
|---|---|---|
Primary | Sequence of amino acids | Peptide bonds |
Secondary | Local folding (α-helix, β-sheet) | Hydrogen bonds (backbone) |
Tertiary | 3D shape of a single polypeptide | Hydrophobic, ionic, H-bonds, disulfide bridges |
Quaternary | Assembly of multiple polypeptides | Same as tertiary, between subunits |
Key Equations and Concepts
Number of possible codons:
Central Dogma:
Additional info: Some genes encode functional RNAs (e.g., rRNA, tRNA) that are not translated into proteins. The central dogma is a general framework, but exceptions exist (e.g., reverse transcription in retroviruses).
