 Back
BackMacromolecules: Carbohydrates – Structure, Function, and Biological Importance
Study Guide - Smart Notes

Macromolecules: An Introduction
Overview of Biological Macromolecules
Biological macromolecules are large, complex molecules essential for life. They include carbohydrates, lipids, proteins, and nucleic acids. Each type of macromolecule plays a unique role in cellular structure and function.
Carbohydrates: Serve as energy sources and structural components.
Lipids: Store energy and form biological membranes.
Proteins: Perform a wide range of functions, including catalysis, structure, and signaling.
Nucleic Acids: Store and transmit genetic information.

Water Properties Review
Significance of Water in Biology
Water is vital for all living organisms due to its unique chemical properties. These properties arise from its polar nature and ability to form hydrogen bonds, making it an excellent solvent and participant in biochemical reactions.
Cohesion and Adhesion: Water molecules stick to each other and to other substances, facilitating transport in plants.
High Specific Heat: Water resists temperature changes, helping organisms maintain stable internal conditions.
Solvent Properties: Water dissolves many substances, enabling chemical reactions in cells.
Ice Floats: Solid water is less dense than liquid water, protecting aquatic life in cold climates.
Monomers and Polymers
Building Blocks of Macromolecules
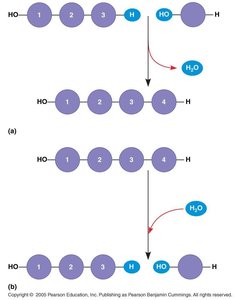
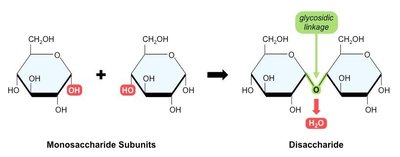
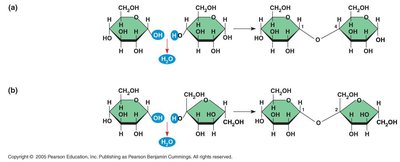
Macromolecules are formed by linking smaller units called monomers into long chains known as polymers. The process of joining monomers is called dehydration synthesis, while breaking polymers into monomers is called hydrolysis.
Monomer: A small molecule that can join with others to form a polymer.
Polymer: A large molecule composed of repeating monomer units.
Dehydration Synthesis: Removal of a water molecule to form a new bond between monomers.
Hydrolysis: Addition of water to break a bond in a polymer, releasing monomers.
Carbohydrates
Functions of Carbohydrates
Carbohydrates are essential biomolecules that serve as energy sources, energy storage, and structural components in living organisms.
Energy Source: Glucose is required for cellular respiration to produce ATP, the energy currency of the cell.
Energy Storage: Glycogen stores energy in animal livers; starch stores energy in plant cells.
Structural Roles: Chitin forms exoskeletons in arthropods; cellulose provides rigidity to plant cell walls and acts as dietary fiber in humans.
Component of Nucleic Acids: Ribose and deoxyribose sugars are part of RNA and DNA, respectively.

Classification of Carbohydrates
Monosaccharides, Disaccharides, Oligosaccharides, and Polysaccharides
Carbohydrates are classified based on the number of sugar units they contain:
Monosaccharides: Single sugar units (e.g., glucose, fructose, galactose).
Disaccharides: Two monosaccharides joined by a glycosidic bond (e.g., sucrose, maltose, lactose).
Oligosaccharides: Chains of 3–9 monosaccharides, often found on cell membranes as recognition molecules.
Polysaccharides: Long chains (10 or more) of monosaccharides (e.g., starch, glycogen, cellulose, chitin).
Monosaccharides
Monosaccharides are the simplest carbohydrates, typically with the formula C6H12O6. They are classified by the number of carbon atoms and the arrangement of atoms in the ring structure.
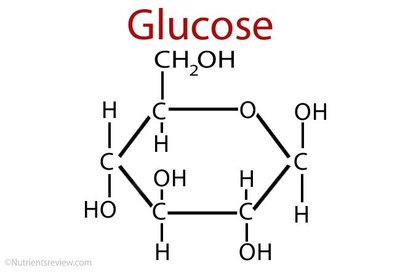
Glucose: Main energy source for cells.
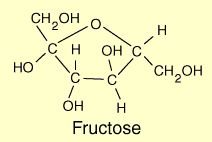
Fructose: Found in fruits and honey.
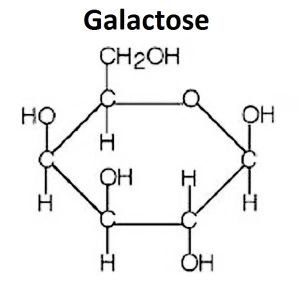
Galactose: Component of lactose in milk.
Disaccharides
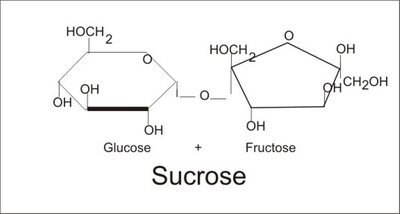
Disaccharides are formed by joining two monosaccharides via a dehydration reaction, creating a glycosidic linkage. Common examples include:
Sucrose: Glucose + Fructose (table sugar)
Maltose: Glucose + Glucose (product of starch digestion)
Lactose: Glucose + Galactose (milk sugar)
Oligosaccharides
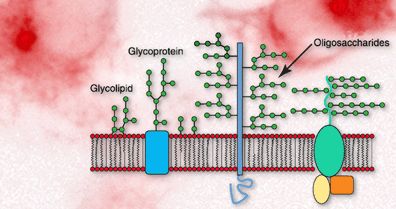
Oligosaccharides are short chains of 3–9 monosaccharide units. They are commonly found attached to proteins and lipids on the cell membrane, playing key roles in cell recognition and signaling.
Function: Cell-cell recognition, immune response, and signaling.
Polysaccharides
Polysaccharides are complex carbohydrates composed of many monosaccharide units. They serve as energy storage or structural materials.
Starch (Amylose): Energy storage in plants.
Glycogen: Energy storage in animals, mainly in the liver and muscles.
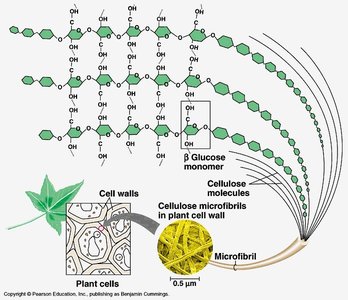
Cellulose: Structural component of plant cell walls; indigestible by humans but important as dietary fiber.
Chitin: Structural component in fungal cell walls and arthropod exoskeletons.
Making and Breaking Carbohydrate Polymers
Dehydration Synthesis and Hydrolysis
Carbohydrate polymers are formed and broken down by specific chemical reactions:
Dehydration Synthesis: Joins two monosaccharides, releasing a water molecule and forming a glycosidic bond.
Hydrolysis: Breaks a glycosidic bond by adding water, splitting a disaccharide or polysaccharide into smaller units.

Biological Importance and Dietary Sources of Carbohydrates
Types of Carbohydrates in the Diet
Carbohydrates in the human diet are classified as sugars, starches, and fiber, each with different effects on metabolism and health.
Type | Description | Examples |
|---|---|---|
Sugar | Quickly converted into energy | Soda, cookies, cereal, juice, treats |
Starches | Takes longer to convert into energy | Bread, pasta, veggies, rice, fruits |
Fiber | Cannot be digested or used for energy | Beans, legumes, nuts, seeds |

Are All Carbohydrates Created Equal?
Although all carbohydrates are eventually broken down into glucose, the source and complexity of the carbohydrate affect digestion, absorption, and health outcomes. Whole foods like fruits and whole grains provide fiber, vitamins, and minerals, while refined sugars offer quick energy but little nutritional value.
Simple Carbohydrates: Rapidly digested and absorbed, causing quick spikes in blood glucose.
Complex Carbohydrates: Digested more slowly, providing sustained energy and additional nutrients.

Summary Table: Carbohydrate Types and Functions
Type | Monomer | Polymer | Function | Example |
|---|---|---|---|---|
Monosaccharide | Glucose, Fructose, Galactose | — | Immediate energy | Glucose in blood |
Disaccharide | Glucose + Fructose, etc. | Sucrose, Maltose, Lactose | Transport, energy | Table sugar (sucrose) |
Oligosaccharide | 3–9 monosaccharides | Oligosaccharides | Cell recognition | Glycoproteins |
Polysaccharide | 10+ monosaccharides | Starch, Glycogen, Cellulose, Chitin | Energy storage, structure | Plant starch, animal glycogen |
Additional info: This guide expands on the original notes by providing definitions, examples, and academic context for each carbohydrate type, as well as relevant chemical equations and dietary implications.
