 Back
BackMembrane Transport and Metabolism in Cells
Study Guide - Smart Notes

Water Balance in Plant Cells
Osmosis and Turgor Pressure
Plant cells maintain water balance through osmosis, which is the movement of water across a selectively permeable membrane. The cell wall provides structural support and helps regulate water uptake.
Turgor Pressure: When a plant cell is in a hypotonic solution, water enters the cell, causing it to swell. The inelastic cell wall exerts back pressure, known as turgor pressure, making the cell turgid (firm), which is the healthy state for most plant cells.
Flaccid Cells: In an isotonic environment, there is no net water movement, and plant cells become flaccid, leading to wilting.
Plasmolysis: In a hypertonic environment, plant cells lose water, causing the membrane to pull away from the cell wall (plasmolysis), which can result in wilting and cell death.
Membrane Transport Mechanisms
Facilitated Diffusion and Channel Proteins
Facilitated diffusion is a passive transport process where specific molecules or ions move across the membrane via channel or carrier proteins, down their concentration gradient, without energy input.
Channel Proteins: Provide corridors for specific molecules or ions (e.g., aquaporins for water, ion channels for ions).
Gated Channels: Some channels open or close in response to stimuli, such as voltage changes or ligand binding.
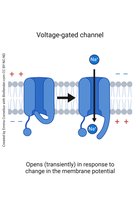
Active Transport
Active transport moves substances against their concentration gradients using energy, typically from ATP hydrolysis. All proteins involved in active transport are carrier proteins.
Primary Active Transport: Direct use of ATP to transport molecules (e.g., sodium-potassium pump).
Secondary Active Transport (Cotransport): Uses the energy from the downhill movement of one solute to drive the uphill movement of another.
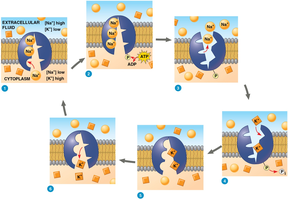
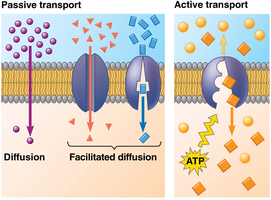
Electrochemical Gradients and Membrane Potential
The membrane potential is the voltage difference across a membrane, created by the unequal distribution of ions. This potential influences the movement of ions, contributing to the electrochemical gradient, which combines both the chemical (concentration) and electrical (charge) forces.
Electrochemical Gradient: Drives the diffusion of ions across membranes.
Membrane Potential: Favors passive transport of cations into and anions out of the cell.
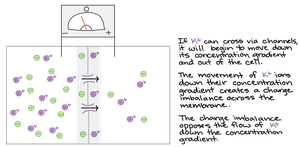
Bulk Transport: Endocytosis
Large molecules, such as proteins and polysaccharides, cross the membrane in bulk via vesicles. Endocytosis is the process by which cells take in substances by engulfing them in vesicles.
Phagocytosis: "Cellular eating"; the cell engulfs large particles using pseudopodia.
Pinocytosis: "Cellular drinking"; the cell takes in extracellular fluid and dissolved solutes in small vesicles.
Receptor-Mediated Endocytosis: Specific molecules bind to receptors, triggering vesicle formation.
Example: Human cells use receptor-mediated endocytosis to take in cholesterol via LDL particles. Defects in this process can lead to familial hypercholesterolemia and increased cardiac risk.
Metabolism and Thermodynamics
Metabolic Pathways
Metabolism encompasses all chemical reactions in an organism, organized into metabolic pathways. Each step is catalyzed by a specific enzyme.
Catabolic Pathways: Break down complex molecules, releasing energy (e.g., cellular respiration).
Anabolic Pathways: Build complex molecules from simpler ones, consuming energy (e.g., protein synthesis).
Bioenergetics: The study of how energy flows through living organisms.
Forms and Conversions of Energy
Energy is the capacity to do work and exists in various forms, such as kinetic, potential, and chemical energy. Living cells transform energy to perform work.
Energy Conversion Example: Chemical energy from food is converted to kinetic energy for movement and potential energy when climbing.
Laws of Thermodynamics in Biology
Thermodynamics studies energy transformations. Biological systems are open systems, exchanging energy and matter with their surroundings.
First Law (Conservation of Energy): Energy can be transferred and transformed, but not created or destroyed.
Second Law: Every energy transfer increases the entropy (disorder) of the universe. Some energy is lost as heat.
Order, Disorder, and Entropy
Living organisms create order locally but increase the overall disorder of their surroundings through metabolic processes. Spontaneous processes increase entropy and can occur without energy input.
Gibbs Free Energy and Spontaneity
Free energy (G) is the portion of a system's energy available to do work. The change in free energy () determines whether a process is spontaneous.
Spontaneous Processes: ; decrease free energy and can be harnessed to do work.
Nonspontaneous Processes: ; require energy input.
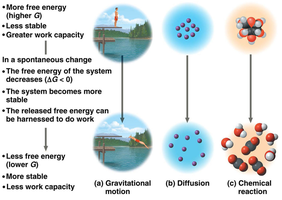
Exergonic Reactions: Release free energy (e.g., cellular respiration). Endergonic Reactions: Absorb free energy (e.g., photosynthesis).
ATP and Cellular Work
ATP Hydrolysis and Energy Coupling
ATP (adenosine triphosphate) is the cell's main energy currency. Hydrolysis of ATP releases energy by breaking the terminal phosphate bond, which is used to power cellular work.
Phosphorylation: Transfer of a phosphate group from ATP to another molecule, making it more reactive.
ATP Cycle: ATP is regenerated from ADP and inorganic phosphate using energy from catabolic reactions.
Types of Cellular Work
Chemical Work: Driving endergonic reactions.
Transport Work: Pumping substances across membranes.
Mechanical Work: Movement, such as muscle contraction or cilia beating.
Enzymes and Reaction Rates
Enzyme Function and Catalysis
Enzymes are biological catalysts that speed up chemical reactions by lowering the activation energy required. They are highly specific for their substrates.
Substrate: The reactant an enzyme acts on.
Active Site: The region on the enzyme where the substrate binds.
Enzyme-Substrate Complex: Temporary complex formed during catalysis.
Factors Affecting Enzyme Activity
Temperature: Each enzyme has an optimal temperature for activity.
pH: Each enzyme has an optimal pH (e.g., pepsin in the stomach at pH 2, trypsin in the intestine at pH 8).
Cofactors: Nonprotein helpers (inorganic ions or organic coenzymes) required for enzyme activity.
Inhibitors: Chemicals that reduce enzyme activity; can be reversible or irreversible.
Activation Energy and Transition State
Activation energy () is the initial energy needed to start a reaction. Enzymes lower $E_A$, allowing reactions to proceed rapidly at cellular temperatures.
Type of Inhibition | Description | Example |
|---|---|---|
Irreversible | Inhibitor covalently binds to enzyme, permanently inactivating it | Penicillin |
Reversible | Inhibitor binds weakly, can dissociate | Many metabolic regulators |
Example: The enzyme sucrase catalyzes the hydrolysis of sucrose to glucose and fructose, a reaction that would otherwise occur extremely slowly at room temperature.
