 Back
BackThe Four Major Biomolecules: Structure, Function, and Biological Importance
Study Guide - Smart Notes

Biomolecules: The Four Major Macromolecules
Introduction to Biomolecules
Biomolecules, also known as macromolecules, are large, complex molecules that are fundamental to the structure and function of living organisms. The four major classes of biomolecules are proteins, nucleic acids, carbohydrates, and lipids. Each class has unique structures and functions, but all are primarily carbon-based, allowing for a diversity of molecular shapes and chemical properties.
Proteins: Polymers of amino acids with diverse cellular functions.
Nucleic Acids: Polymers of nucleotides responsible for information storage and transfer.
Carbohydrates: Sugars and their polymers, serving as energy sources and structural components.
Lipids: Hydrophobic molecules including fats, phospholipids, and steroids, important for energy storage and membrane structure.
Carbon: The Foundation of Biomolecules
Properties of Carbon
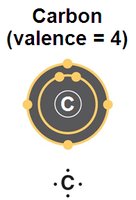
Carbon atoms are the backbone of all biomolecules due to their ability to form four covalent bonds, resulting in a variety of stable structures such as chains, rings, and branches. This versatility enables the complexity and diversity of organic molecules found in living systems.
Valence of 4: Carbon can form four covalent bonds with other atoms, including hydrogen, oxygen, and nitrogen.
Structural Diversity: Carbon's bonding properties allow for the formation of straight chains, branched chains, and rings.
Polymers and Monomers
Formation and Breakdown of Polymers
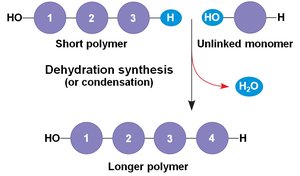
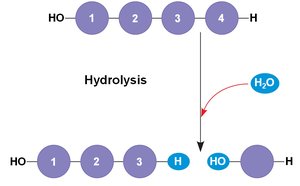
Most biomolecules are polymers, formed by linking smaller units called monomers. The process of joining monomers is called dehydration synthesis (or condensation), while the breakdown of polymers into monomers occurs via hydrolysis.
Dehydration Synthesis: Removes a water molecule to form a new bond between monomers.
Hydrolysis: Adds a water molecule to break a bond between monomers.
Carbohydrates
Structure and Function
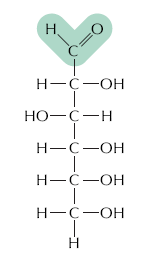
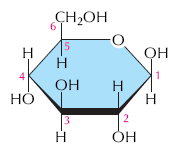
Carbohydrates are organic molecules with the general formula (CH2O)n. They serve as energy sources and structural materials in cells. The simplest carbohydrates are monosaccharides (e.g., glucose), which can be joined to form disaccharides and polysaccharides (e.g., starch, glycogen, cellulose).
Monosaccharides: Simple sugars, such as glucose (C6H12O6), that are the primary energy source for cells.
Disaccharides: Two monosaccharides joined by a glycosidic bond (e.g., sucrose).
Polysaccharides: Long chains of monosaccharides; serve as energy storage (starch, glycogen) or structural support (cellulose).
Lipids
Types and Functions
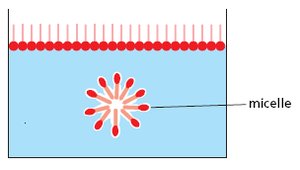
Lipids are a diverse group of hydrophobic molecules that include fats, phospholipids, and steroids. They are not true polymers but are assembled from smaller components. Lipids are essential for energy storage, membrane structure, and signaling.
Fats (Triglycerides): Composed of glycerol and three fatty acids; store energy efficiently.
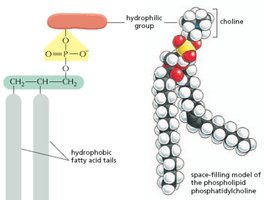
Phospholipids: Major components of cell membranes, consisting of a glycerol backbone, two fatty acids, and a phosphate group.
Steroids: Characterized by four fused carbon rings; include hormones like cholesterol.
Proteins
Structure and Function
Proteins are polymers of amino acids and perform a vast array of functions in cells, including catalysis (enzymes), transport, structure, signaling, and defense. The function of a protein is determined by its three-dimensional shape, which is specified by the sequence of amino acids.
Amino Acids: The building blocks of proteins; 20 different types exist, each with a unique side chain (R-group).
Peptide Bonds: Covalent bonds that link amino acids together in a polypeptide chain.
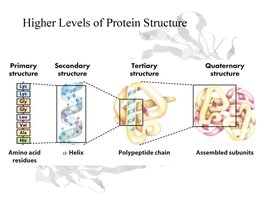
Protein Structure: Defined at four levels—primary, secondary, tertiary, and quaternary.
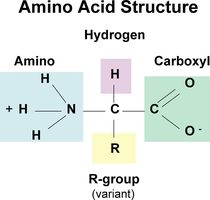
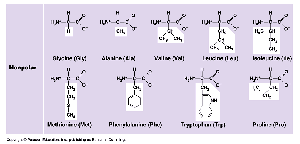
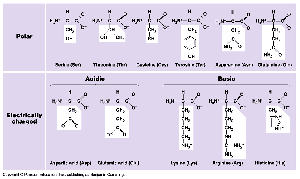
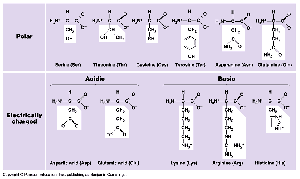
Levels of Protein Structure
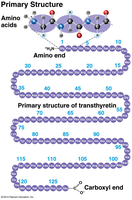
Primary Structure: The linear sequence of amino acids in a polypeptide.
Secondary Structure: Local folding into alpha-helices and beta-sheets, stabilized by hydrogen bonds.
Tertiary Structure: The overall three-dimensional shape of a polypeptide, determined by interactions among R-groups.
Quaternary Structure: The association of multiple polypeptide chains into a functional protein (only in some proteins).
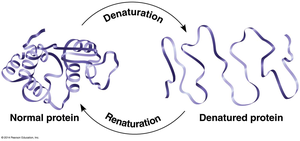
Protein Denaturation
Protein structure is sensitive to environmental conditions such as temperature, pH, and chemicals. Denaturation is the process by which a protein loses its native shape and, consequently, its function.
Denatured Protein: Unfolded and nonfunctional.
Renaturation: Some proteins can refold and regain function if normal conditions are restored.

Nucleic Acids
Structure and Function
Nucleic acids are polymers of nucleotides and are responsible for the storage, transmission, and expression of genetic information. The two main types are DNA (deoxyribonucleic acid) and RNA (ribonucleic acid).
DNA: Stores genetic information; double-stranded helix.
RNA: Transfers genetic information for protein synthesis; usually single-stranded.
ATP: A nucleotide that acts as the primary energy carrier in cells.
cAMP: A nucleotide involved in cell signaling.
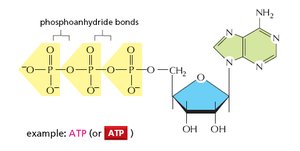
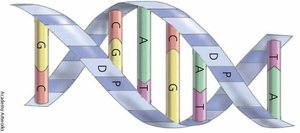
Nucleotide Structure
Each nucleotide consists of three components:
Phosphate group
Pentose sugar (deoxyribose in DNA, ribose in RNA)
Nitrogenous base (adenine, guanine, cytosine, thymine in DNA; uracil replaces thymine in RNA)
DNA vs. RNA
Feature | DNA | RNA |
|---|---|---|
Sugar | Deoxyribose | Ribose |
Bases | A, T, G, C | A, U, G, C |
Strands | Double-stranded | Single-stranded |
Summary Table: The Four Major Biomolecules
Biomolecule | Monomer | Polymer | Main Functions |
|---|---|---|---|
Carbohydrates | Monosaccharide | Polysaccharide | Energy, structure |
Lipids | Fatty acids, glycerol | Not true polymers | Energy storage, membranes, signaling |
Proteins | Amino acid | Polypeptide | Catalysis, structure, transport, signaling |
Nucleic Acids | Nucleotide | DNA, RNA | Information storage and transfer |
