 Back
BackThe Structure and Function of Large Biological Molecules
Study Guide - Smart Notes

The Structure and Function of Large Biological Molecules
Introduction to Macromolecules
All living organisms are composed of four major classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic acids. These macromolecules are essential for life and exhibit unique properties due to the specific arrangement of their atoms.
Macromolecules are large, complex molecules.
They are built from smaller units called monomers.
Carbohydrates, proteins, and nucleic acids are polymers, meaning they are composed of repeating monomer units.

Polymer Synthesis and Breakdown
Polymers are synthesized and broken down by specific chemical reactions:
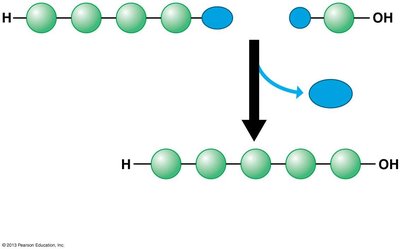
Dehydration reaction: Joins two monomers by removing a water molecule, forming a covalent bond.
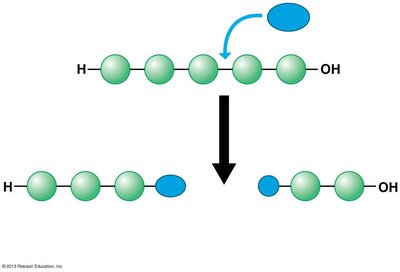
Hydrolysis: Breaks a polymer into monomers by adding a water molecule, reversing the dehydration reaction.
Enzymes catalyze both synthesis and breakdown of polymers.

Carbohydrates: Fuel and Building Material
Monosaccharides and Disaccharides
Carbohydrates include sugars and their polymers. The simplest carbohydrates are monosaccharides (simple sugars), which serve as major fuel for cells and as raw material for building other molecules.
General formula:
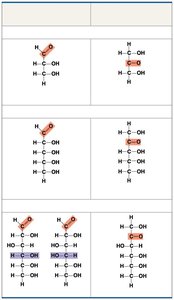
Classified by the location of the carbonyl group (aldose or ketose) and the number of carbons in the skeleton (triose, pentose, hexose).
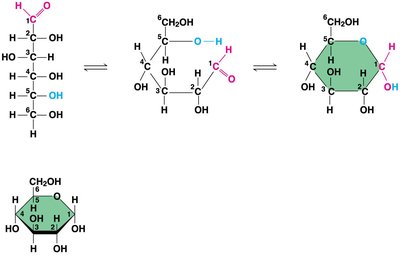
Monosaccharides often form rings in aqueous solutions.
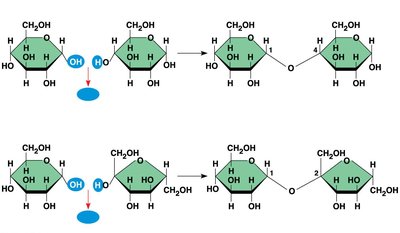
Disaccharides are formed by joining two monosaccharides via a glycosidic linkage (covalent bond formed by a dehydration reaction).
Polysaccharides: Storage and Structure
Polysaccharides are polymers of sugars and serve storage or structural roles. Their function is determined by the type of monomers and the glycosidic linkages between them.
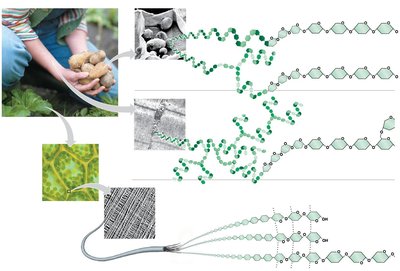
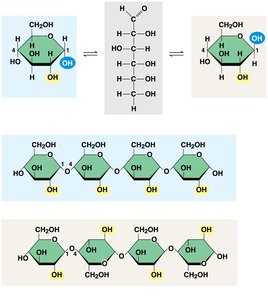
Starch: Storage polysaccharide in plants, composed of glucose monomers (amylose and amylopectin).
Glycogen: Storage polysaccharide in animals, stored mainly in liver and muscle cells.
Cellulose: Structural polysaccharide in plant cell walls, composed of β-glucose monomers.
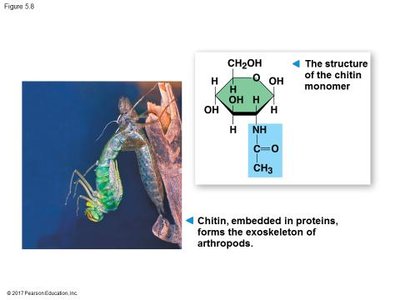
Chitin: Structural polysaccharide in the exoskeleton of arthropods and cell walls of fungi.
Lipids: Hydrophobic Molecules
Fats
Lipids are a diverse group of hydrophobic molecules that do not form true polymers. The most important lipids are fats, phospholipids, and steroids.
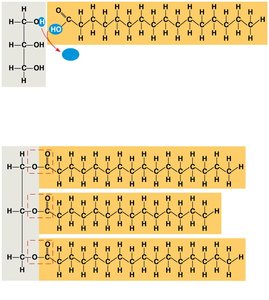
Fats are constructed from glycerol and fatty acids.
Glycerol: Three-carbon alcohol with a hydroxyl group on each carbon.
Fatty acid: Carboxyl group attached to a long hydrocarbon chain.
Fats are formed by dehydration reactions, creating ester linkages.
Saturated fatty acids: No double bonds, maximum hydrogen atoms, solid at room temperature.
Unsaturated fatty acids: One or more double bonds, liquid at room temperature, can be cis or trans (trans fats).
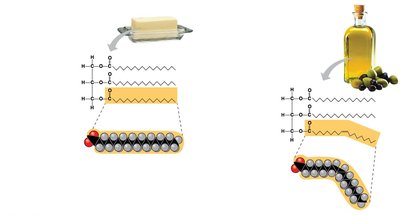
Fats function as energy storage, insulation, and cushioning for organs.
Phospholipids
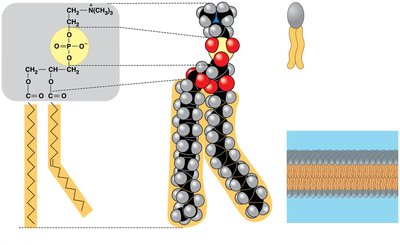
Phospholipids consist of two fatty acids and a phosphate group attached to glycerol. They are amphipathic, with hydrophobic tails and a hydrophilic head, and form the basis of cell membranes.
Steroids
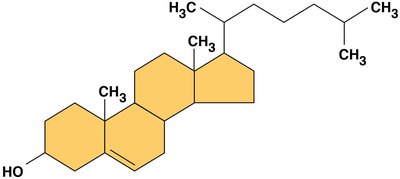
Steroids are lipids with a carbon skeleton of four fused rings. Cholesterol is an important steroid in animal cell membranes and a precursor for other steroids.
Proteins: Structure and Function
Protein Functions
Proteins are the most diverse macromolecules, accounting for more than 50% of the dry mass of most cells. They perform a wide range of functions, including catalysis, defense, storage, transport, communication, movement, and structural support.
Amino Acids and Polypeptides
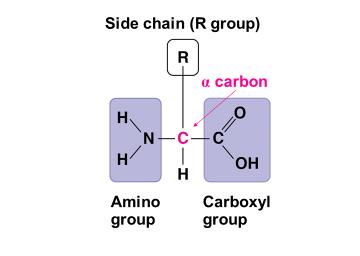
Proteins are polymers of amino acids, which have a central carbon (α carbon) bonded to an amino group, a carboxyl group, a hydrogen atom, and a variable side chain (R group).
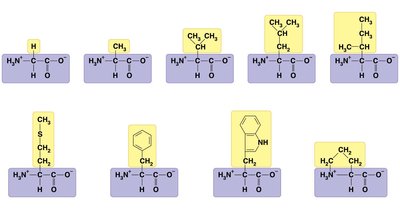
Amino acids are classified by the properties of their side chains: nonpolar (hydrophobic), polar (hydrophilic), acidic (negatively charged), or basic (positively charged).
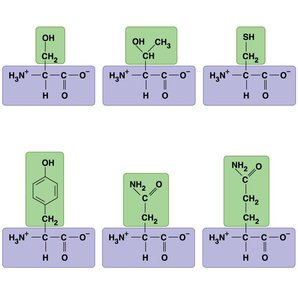
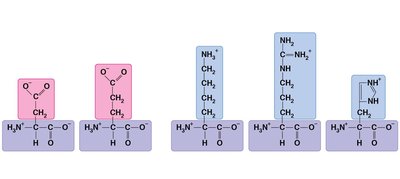
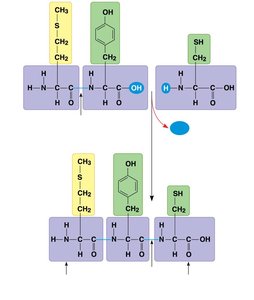
Amino acids are linked by peptide bonds to form polypeptides.
Each polypeptide has an amino (N) terminus and a carboxyl (C) terminus.
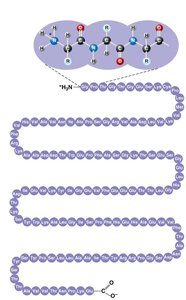
Levels of Protein Structure
Primary structure: Unique sequence of amino acids.
Secondary structure: Coils (α helix) and folds (β pleated sheet) stabilized by hydrogen bonds.
Tertiary structure: Overall 3D shape due to interactions among R groups (hydrogen bonds, ionic bonds, hydrophobic interactions, disulfide bridges).
Quaternary structure: Association of multiple polypeptide chains.
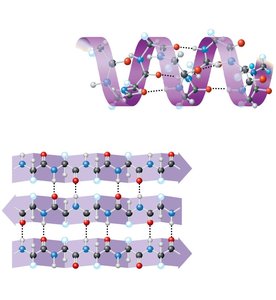
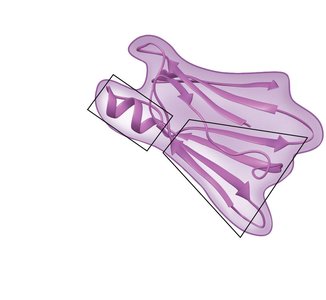
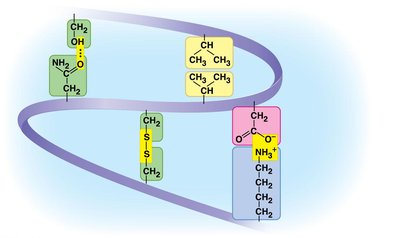
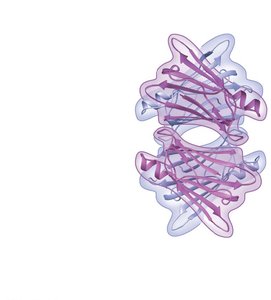

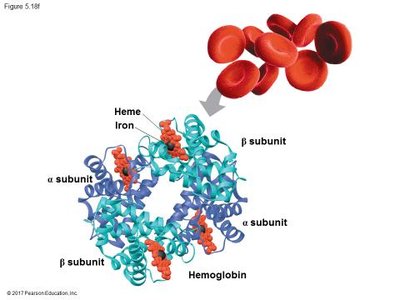
Protein Folding and Denaturation
The function of a protein depends on its specific shape, which is determined by its amino acid sequence and environmental conditions. Changes in pH, temperature, or salt concentration can cause denaturation, leading to loss of function.
Nucleic Acids: Information Molecules
DNA and RNA
Nucleic acids store, transmit, and help express hereditary information. The two types are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
Genes are units of inheritance made of DNA.
DNA directs its own replication and the synthesis of RNA (gene expression: DNA → RNA → protein).
Nucleotide Structure
Nucleic acids are polymers called polynucleotides, made of nucleotide monomers.
Each nucleotide consists of a nitrogenous base, a pentose sugar, and one or more phosphate groups.
Nitrogenous bases: pyrimidines (C, T, U) and purines (A, G).
Sugars: deoxyribose (DNA) and ribose (RNA).
DNA and RNA Structure
DNA: Double helix, antiparallel strands, complementary base pairing (A=T, C≡G).
RNA: Single-stranded, can form complex shapes by internal base pairing, uracil (U) replaces thymine (T).
Genomics and Proteomics
Modern Biological Inquiry
Advances in sequencing technology have enabled the study of entire genomes (genomics) and large sets of proteins (proteomics). These fields use computational tools (bioinformatics) to analyze large datasets, revolutionizing our understanding of biology, evolution, and disease.
Summary Table: Major Classes of Large Biological Molecules
Class | Monomer | Polymer | Bond Type | Main Functions |
|---|---|---|---|---|
Carbohydrates | Monosaccharide | Polysaccharide | Glycosidic linkage | Energy storage, structure |
Lipids | Fatty acids, glycerol | Not true polymers | Ester linkage | Energy storage, membranes, signaling |
Proteins | Amino acid | Polypeptide | Peptide bond | Catalysis, structure, transport, etc. |
Nucleic Acids | Nucleotide | Polynucleotide | Phosphodiester bond | Information storage and transfer |
