 Back
BackThe Structure and Function of Large Biological Molecules
Study Guide - Smart Notes

Overview: The Molecules of Life
Introduction to Biological Macromolecules
All living organisms are composed of four major classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic acids. These molecules are known as organic compounds because they contain carbon atoms.
Macromolecules are large molecules made up of thousands of covalently bonded atoms.
Carbohydrates, proteins, and nucleic acids are considered macromolecules.
Macromolecules as Polymers
Polymers and Monomers
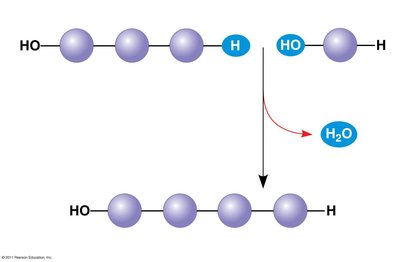
A polymer is a long molecule consisting of many similar or identical building blocks called monomers.
Three classes of life's organic molecules are polymers: carbohydrates, proteins, and nucleic acids.
Synthesis and Breakdown of Polymers
Dehydration reaction (condensation): Joins two monomers by removing a water molecule.
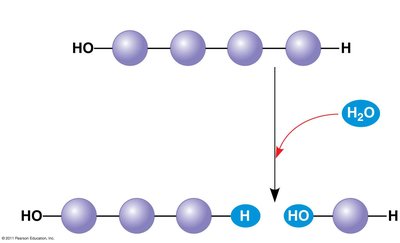
Hydrolysis: Breaks polymers into monomers by adding a water molecule, essentially the reverse of dehydration.
Carbohydrates
Structure and Function
Carbohydrates include sugars and polymers of sugars.
Function: Serve as the primary source of energy and as structural materials.
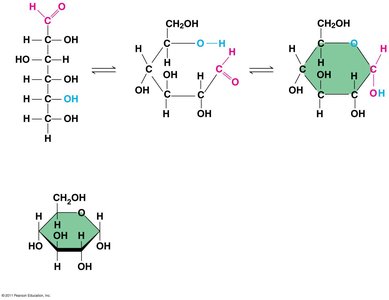
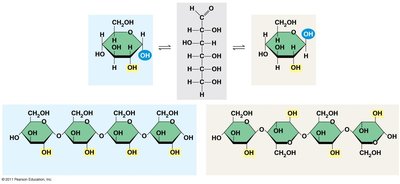
The simplest carbohydrates are monosaccharides (single sugars).
Carbohydrate macromolecules are polysaccharides, polymers composed of many sugar monomers.

Monosaccharides
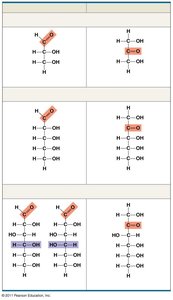
General formula: multiples of CH2O.
Glucose (C6H12O6) is the most common monosaccharide.
Classified by the location of the carbonyl group (aldose or ketose) and the number of carbons.
Ring Formation
In aqueous solutions, many sugars form ring structures.
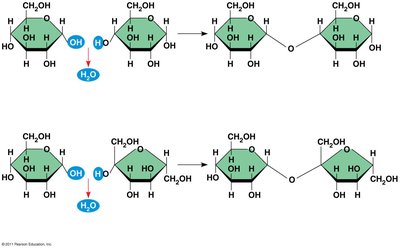
Disaccharides
Formed by a dehydration reaction joining two monosaccharides.
The covalent bond is called a glycosidic linkage.
Examples:
Glucose + Glucose = Maltose
Glucose + Fructose = Sucrose
Glucose + Galactose = Lactose
Polysaccharides
Polysaccharides have storage and structural roles.
The structure and function depend on the sugar monomers and glycosidic linkages.
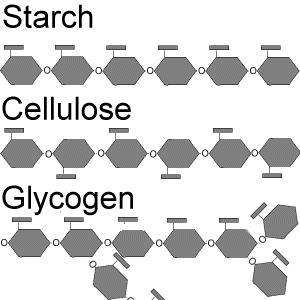
Storage Polysaccharides
Starch: Storage polysaccharide in plants, composed entirely of glucose monomers.
Glycogen: Storage polysaccharide in animals, stored mainly in liver and muscle cells.
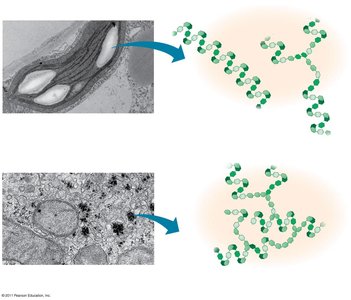
Structural Polysaccharides
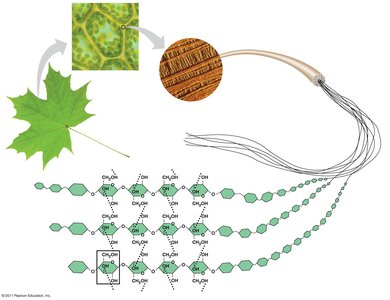
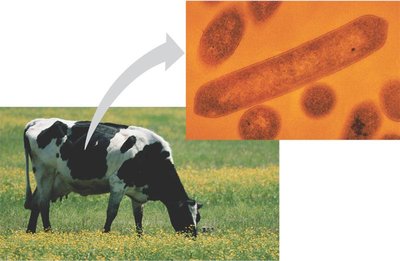
Cellulose: Major component of plant cell walls; polymer of glucose with beta glycosidic linkages.
Cellulose is difficult to digest; only certain microbes can break it down.
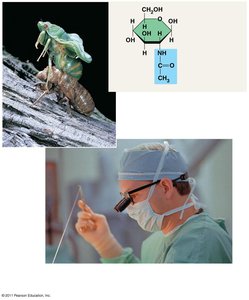
Chitin
Chitin: Structural polysaccharide found in the exoskeleton of arthropods and cell walls of fungi.
Used in surgical thread due to its strength and biodegradability.
Lipids
Structure and Function
Lipids are hydrophobic molecules that do not form polymers.
Consist mostly of hydrocarbons, forming nonpolar covalent bonds.
Major types: fats, phospholipids, and steroids.

Fats
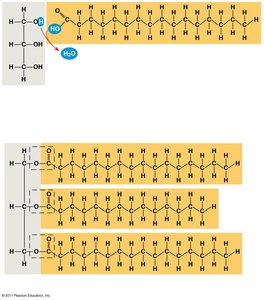
Constructed from glycerol and fatty acids.
Glycerol: Three-carbon alcohol with hydroxyl groups.
Fatty acid: Carboxyl group attached to a long carbon skeleton.
Three fatty acids joined to glycerol by ester linkage form a triglyceride.
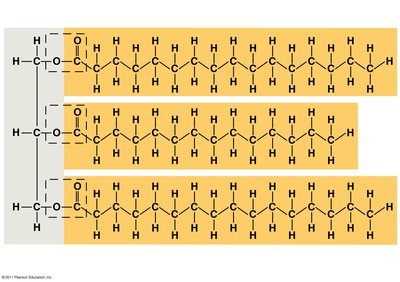
Saturated vs. Unsaturated Fatty Acids
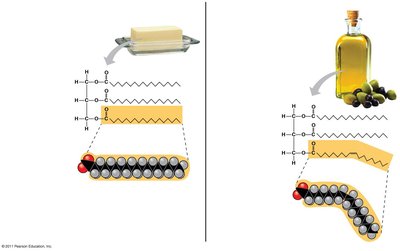
Saturated fatty acids: Maximum hydrogen atoms, no double bonds, solid at room temperature.
Unsaturated fatty acids: One or more double bonds, liquid at room temperature.
Trans fats are produced by hydrogenation and may contribute to cardiovascular disease.
Essential fatty acids (e.g., omega-3) must be obtained from diet.
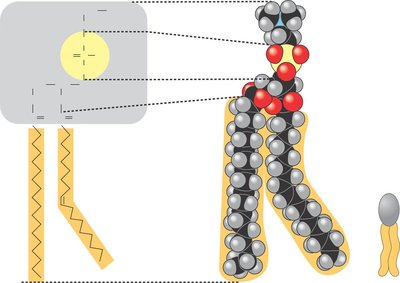
Phospholipids
Two fatty acids and a phosphate group attached to glycerol.
Hydrophilic head and hydrophobic tails.
Form bilayers in water, major component of cell membranes.
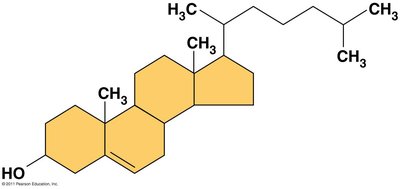
Steroids
Steroids: Lipids with a carbon skeleton of four fused rings.
Cholesterol: Essential in animal cell membranes, but high levels can cause disease.
Proteins
Structure and Function
Proteins account for more than 50% of the dry mass of most cells.
Functions: Structural support, storage, transport, communication, movement, defense.

Polypeptides and Amino Acids
Proteins are made of one or more polypeptides, polymers of amino acids.
Amino acids have carboxyl and amino groups, differing by their side chains (R groups).
Linked by peptide bonds.
Protein Structure
Four levels: primary (sequence), secondary (alpha helix, beta sheet), tertiary (3D folding), quaternary (multiple polypeptides).
Structure determines function.
Sickle-Cell Disease
A single amino acid substitution in hemoglobin causes sickle-cell disease, affecting protein function and cell shape.
Protein Denaturation and Folding
Physical and chemical conditions (pH, salt, temperature) can cause proteins to denature (lose structure and function).
Chaperonins assist in proper protein folding; misfolded proteins are linked to diseases.
Enzymes
Enzyme Function
Enzymes are proteins that act as catalysts, speeding up biochemical reactions by lowering activation energy.
Each enzyme is specific to its substrate and reaction.
Enzyme names often end in -ase.
Enzyme Vocabulary
Enzyme: Helper protein molecule
Substrate: Molecule acted upon by enzyme
Products: Result of enzyme-catalyzed reaction
Active site: Region of enzyme where substrate binds
Nucleic Acids
Structure and Function
Nucleic acids store, transmit, and help express hereditary information.
Genes are made of DNA, a nucleic acid composed of nucleotides.
Two types: DNA (deoxyribonucleic acid) and RNA (ribonucleic acid).
DNA directs its own replication and protein synthesis via mRNA.
Nucleotide Structure
Nucleic acids are polymers called polynucleotides.
Each nucleotide consists of a nitrogenous base, a pentose sugar, and one or more phosphate groups.
A nucleoside is a nucleotide without the phosphate group.
Macromolecule | Monomer | Polymer | Main Function |
|---|---|---|---|
Carbohydrate | Monosaccharide | Polysaccharide | Energy, structure |
Lipid | Fatty acid, glycerol | Triglyceride, phospholipid, steroid | Energy storage, membranes, hormones |
Protein | Amino acid | Polypeptide | Structure, enzymes, transport, defense |
Nucleic Acid | Nucleotide | DNA, RNA | Genetic information |
Additional info: Table above summarizes the main macromolecules, their monomers, polymers, and functions for clarity.
