 Back
BackThe Structure and Function of Large Biological Molecules
Study Guide - Smart Notes

Chapter 5: The Structure and Function of Large Biological Molecules
Four Major Classes of Biological Molecules
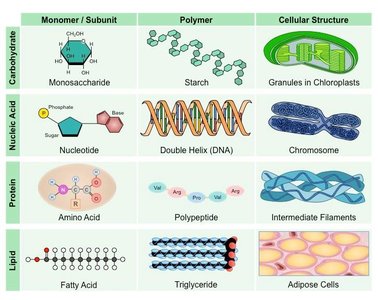
Living organisms are composed of four major classes of large biological molecules, each essential for life. These molecules differ in structure and function but are all crucial for cellular processes.
Carbohydrates: Serve as fuel and building material.
Lipids: Important for energy storage, membrane structure, and signaling.
Proteins: Perform a vast array of functions, including catalysis, structure, and transport.
Nucleic Acids: Store and transmit genetic information.
Monomers and Polymers
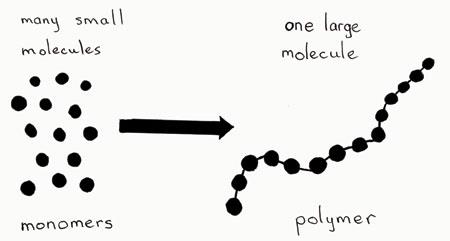
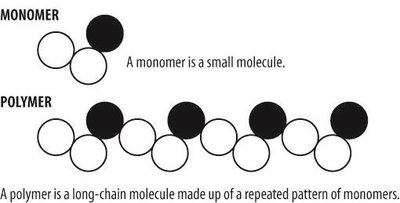
Most biological macromolecules are polymers, long molecules built from repeating units called monomers. The process of linking monomers forms polymers, which can then be organized into complex structures.
Polymer: A long molecule consisting of many similar or identical building blocks linked by covalent bonds.
Monomer: The smaller, repeating molecules that serve as the building blocks of a polymer.
Three of the four classes of life’s organic molecules are polymers: carbohydrates, proteins, and nucleic acids.
Synthesis and Disassembly of Polymers
Polymers are assembled and disassembled by specific chemical reactions, often facilitated by enzymes.
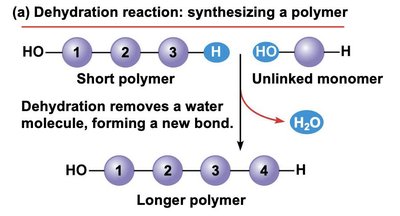
Dehydration Synthesis (Condensation Reaction)
Monomers are joined together by dehydration synthesis, a reaction in which a water molecule is removed to form a new covalent bond.
This process is catalyzed by enzymes in cells.
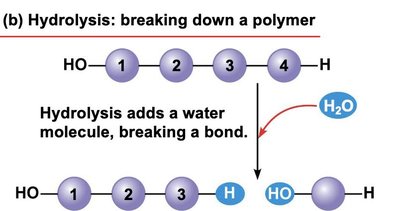
Hydrolysis
Polymers are broken down into monomers by hydrolysis, a reaction in which a water molecule is added, breaking a covalent bond.
Enzymes also facilitate hydrolysis in cells.
Carbohydrates
Overview of Carbohydrates
Carbohydrates are organic molecules composed of carbon, hydrogen, and oxygen, typically in a ratio close to 1:2:1. They serve as energy sources and structural materials in cells.
Include both simple sugars (monosaccharides) and polymers of sugars (polysaccharides).
Monosaccharides
Monosaccharides are the simplest carbohydrates, also known as simple sugars. They are the monomers from which more complex carbohydrates are built.
General formula:
Example: Glucose ()
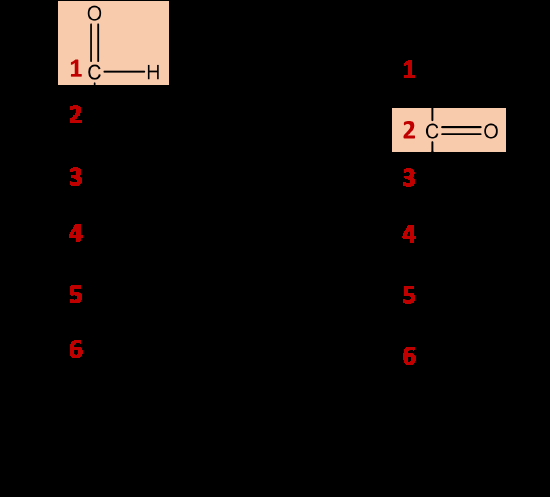
Classified by the location of the carbonyl group:
Aldose: Carbonyl group at the end of the carbon chain.
Ketose: Carbonyl group within the carbon chain.
Classification by Carbon Number
Trioses: 3 carbon atoms (e.g., glyceraldehyde, dihydroxyacetone)
Pentoses: 5 carbon atoms (e.g., ribose, ribulose)
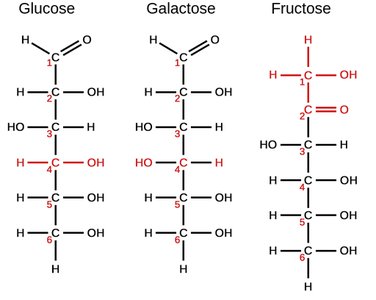
Hexoses: 6 carbon atoms (e.g., glucose, galactose, fructose)
Isomerism in Monosaccharides
Monosaccharides can exist as structural isomers, differing in the arrangement of atoms.
Glucose, galactose, and fructose are all hexoses but differ in structure and properties.
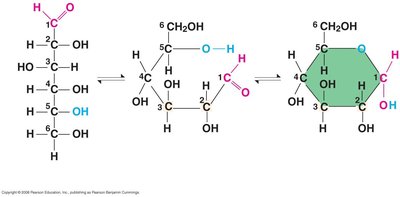
Linear and Ring Forms
Monosaccharides such as glucose can exist in both linear and ring forms in aqueous solutions.
Disaccharides
Disaccharides are formed when two monosaccharides are joined by a dehydration reaction, creating a covalent bond known as a glycosidic linkage.
Glycosidic linkage: The covalent bond formed between two monosaccharides by a dehydration reaction.
Common disaccharides:
Maltose: Glucose + Glucose
Sucrose: Glucose + Fructose
Lactose: Glucose + Galactose
Example: The synthesis of sucrose involves a dehydration reaction between glucose and fructose, forming a 1–2 glycosidic linkage.
