19. Chemical Thermodynamics
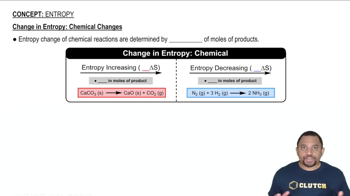
Entropy

Get help from an AI Tutor
Ask a question to get started.
Problem 28b
Textbook Question
Textbook Question(a) What sign for ΔS do you expect when the pressure on 0.600 mol of an ideal gas at 350 K is increased isothermally from an initial pressure of 0.750 atm? (b) If the final pressure on the gas is 1.20 atm, calculate the entropy change for the process. (c) Do you need to specify the temperature to calculate the entropy change?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
542
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos