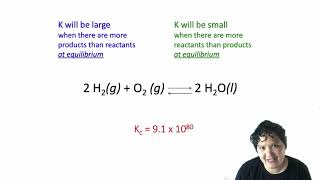16. Chemical Equilibrium
Reaction Quotient
Problem 47
Textbook Question
Textbook QuestionConsider the reaction: NH4HS(s) ⇌ NH3(g) + H2S(g) At a certain temperature, Kc = 8.5 * 10 - 3. A reaction mixture at this temperature containing solid NH4HS has [NH3] = 0.166 M and [H2S] = 0.166 M. Will more of the solid form or will some of the existing solid decompose as equilibrium is reached?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
3622
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos