15. Chemical Kinetics
Integrated Rate Law

Get help from an AI Tutor
Ask a question to get started.
Problem 95
Textbook Question
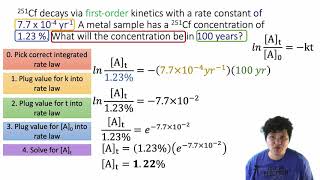
Textbook QuestionThe reaction 2 NO2¡2 NO + O2 has the rate constant k = 0.63 M- 1s - 1. (b) If the initial concentration of NO2 is 0.100 M, how would you determine how long it would take for the concentration to decrease to 0.025 M?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
1087
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos