 Back
BackAcid-Base Equilibria and Polyprotic Acids: Study Notes for General Chemistry
Study Guide - Smart Notes

Acid–Base Equilibria
Introduction to Acid–Base Equilibria
Acid–base equilibria describe the balance between acids and bases in aqueous solutions, focusing on the extent of ionization and the calculation of pH. Understanding these equilibria is essential for predicting the behavior of acids, bases, and their salts in solution.
The pH Scale
pH is a measure of the hydrogen ion concentration in a solution, defined as:
Acidic solutions have pH < 7, neutral solutions have pH = 7, and basic solutions have pH > 7 at 25°C.
Equilibria in Solutions of Weak Acids and Bases
Weak acids and bases do not completely ionize in water. Their equilibria are described by the acid dissociation constant () and base dissociation constant ().
Weak Acid Example:
Weak Base Example:
Calculating pH from or
To determine the pH of a weak acid or base solution, follow these steps:
Write the balanced equation for the principal acid–base reaction.
Set up an ICE (Initial, Change, Equilibrium) table for all species involved.
Substitute equilibrium concentrations into the equilibrium expression and solve for (the change in concentration).
Calculate the concentrations of all species at equilibrium.
Calculate the pH using the equilibrium .

Worked Example: Acetic Acid
Given: ,
Set up the equilibrium:
Let at equilibrium.
Assume is small compared to initial acid concentration: ,
Assumption Check: If is less than 5% of the initial concentration, the assumption is valid. For weak acids with , this is generally safe.
Factors Affecting Acid Strength
Trends in Acid Strength
Across a Period: Acid strength increases with increasing electronegativity of the central atom (e.g., ).
Down a Group: Acid strength increases as the size of the central atom increases, due to decreasing bond strength (e.g., ).
Oxoacids (Oxyacids): For acids with the same number of oxygen atoms, acid strength increases with the electronegativity of the central atom (e.g., ).
For oxoacids with different numbers of oxygen atoms, acid strength increases with the number of oxygen atoms (e.g., ).
Examples and Applications
Predicting Relative Acid Strength:
Between and , $\mathrm{H_2Se}$ is stronger (larger atom).
Between and , $\mathrm{H_2SO_4}$ is stronger (more oxygen atoms).
Between and , $\mathrm{H_2SO_4}$ is stronger (higher oxidation state and more oxygen atoms).
pH of Salt Solutions
Salts from Strong Acids and Bases
Salts formed from strong acids and strong bases (e.g., NaCl, KNO3) do not affect the pH of the solution; their solutions are neutral.
Salts from Weak Acids or Bases
Salts from weak acids and strong bases (e.g., sodium acetate) produce basic solutions because the conjugate base hydrolyzes water to form OH–.
Salts from weak bases and strong acids (e.g., ammonium chloride) produce acidic solutions because the conjugate acid hydrolyzes water to form H3O+.
Highly charged metal cations (e.g., Fe3+, Al3+) act as Lewis acids and can acidify solutions.
Worked Example: pH of KF Solution
Given: , for
Set up equilibrium:
Calculate and then pOH, then pH.
Polyprotic Acids and Bases
Definition and Examples
A polyprotic acid can donate more than one proton (e.g., , , ). A polyprotic base can accept more than one proton (e.g., , ).
Successive Ionization Steps
Each proton is lost in a separate equilibrium step, each with its own value. The first ionization constant () is always larger than the second (), and so on.
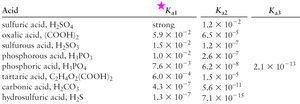
Example:
,
,
For most polyprotic acids, only the first ionization significantly affects pH, except when is not negligible (e.g., ).
Worked Example: pH of 0.10 M
First ionization is complete:
Second ionization: ,
Set up quadratic:
Solve for , ,
pH of Salts of Polyprotic Acids
Estimating pH of Polyprotic Acid Salts
The pH of a solution of a salt of a weak polyprotic acid can be approximated by averaging the relevant p values of the parent acid. This method helps determine whether the solution will be acidic or basic.

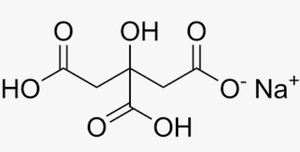
Example: For monosodium citrate (from citric acid, , , ), the average of and estimates the pH.
Worked Example: 1,4-Butanedicarboxylic Acid
Given: ,
In a 0.01 M solution, the species present in the highest concentration is the singly deprotonated form, , due to the relative sizes of and .
Additional info: The structure of monosodium citrate is shown above, which is relevant for understanding the acid–base properties of polyprotic acids and their salts.
