 Back
BackAcid-Base Equilibria: Concepts, Calculations, and Applications
Study Guide - Smart Notes

Acid-Base Equilibria
Introduction to Acid-Base Equilibria
Acid-base equilibria are fundamental to understanding chemical reactions in aqueous solutions. This topic covers the definitions of acids and bases, their behavior in water, and the mathematical relationships used to describe their equilibria.
Brønsted-Lowry and Lewis Definitions
Brønsted-Lowry Acids and Bases
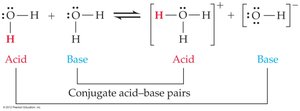
The Brønsted-Lowry definition classifies acids as proton donors and bases as proton acceptors. When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid. - Acid: Donates a proton (H+). - Base: Accepts a proton. - Conjugate acid: The species formed when a base gains a proton. - Conjugate base: The species formed when an acid loses a proton. 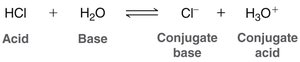
Lewis Acids and Bases
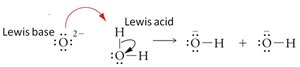
The Lewis definition expands the concept: - Lewis acid: Electron pair acceptor (often lacks a non-bonding electron pair). - Lewis base: Electron pair donor (must have a non-bonding electron pair). 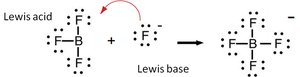
Acids and Bases in Aqueous Solution
Strong and Weak Acids
Acids are classified by their degree of dissociation in water: - Strong acids: Fully dissociate into ions. - Weak acids: Partially dissociate; equilibrium favors reactants. 
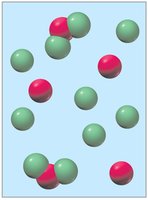
Strong and Weak Bases
Bases are similarly classified: - Strong bases: Completely remove hydrogen from water. - Weak bases: Only partially remove hydrogen; equilibrium favors reactants. 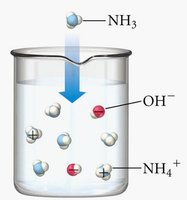
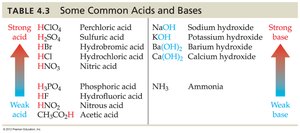
Common Acids and Bases
The strength of acids and bases varies widely. Table 4.3 summarizes common examples:
Strong Acids | Weak Acids | Strong Bases | Weak Bases |
|---|---|---|---|
HClO4, H2SO4, HBr, HCl, HNO3 | H3PO4, HF, HNO2, CH3COOH | NaOH, KOH, Ba(OH)2, Ca(OH)2 | NH3 |
Equilibrium Expressions and Constants
Acid Dissociation Constant (Ka)
The equilibrium constant for acid dissociation is called Ka: - Ka: Indicates acid strength; higher Ka means stronger acid. 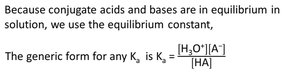

Base Dissociation Constant (Kb)
The equilibrium constant for base dissociation is Kb: - Kb: Indicates base strength; higher Kb means stronger base.
Relationship Between Ka and Kb
For conjugate acid-base pairs:
pKa and pKb
- pKa: - pKb: - Relationship:
Dissociation of Water and the pH Scale
Ion-Product Constant for Water (Kw)
Water self-ionizes to produce hydronium and hydroxide ions: at 25°C. 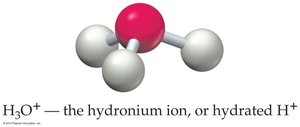
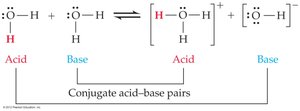
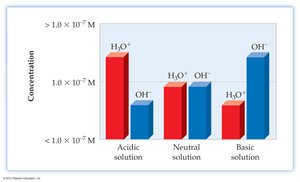
pH and pOH Calculations
- pH: - pOH: - Relationship: - Hydronium ion: H3O+ is the hydrated form of H+. 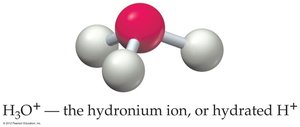
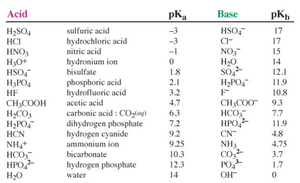
Acid and Base Strength Comparisons
Table of Acid and Base Strengths
Acid and base strengths are often compared using pKa and pKb values. Lower pKa means stronger acid; lower pKb means stronger base.
Acid | pKa | Base | pKb |
|---|---|---|---|
H2SO4 | -3 | HSO4- | 17 |
HCl | -3 | Cl- | 17 |
HNO3 | -1 | NO3- | 15 |
HF | 3.2 | F- | 10.8 |
CH3COOH | 4.7 | CH3COO- | 9.3 |
NH4+ | 9.25 | NH3 | 4.75 |
H2O | 14 | OH- | 1.7 |
Base Strength Table
Base | Kb | pKb |
|---|---|---|
Pyridine, C5H5N | 1.8 × 10-9 | 8.75 |
Hydroxylamine, NH2OH | 1.1 × 10-8 | 7.97 |
Ammonia, NH3 | 1.8 × 10-5 | 4.75 |
Trimethylamine, (CH3)3N | 6.5 × 10-5 | 4.19 |

Equilibrium Calculations and Applications
Predicting Equilibrium Direction
The equilibrium position in acid-base reactions is determined by comparing the strengths of acids and bases involved. - Equilibrium favors the weaker acid/base. - Example: If Ka for H3O+ is much larger than for CH3CO2H, equilibrium favors the side with CH3CO2H.
Calculating pH and Degree of Ionization
- pH calculation: - Degree of ionization:
Example Problems
- Calculate pH given [OH-]: Use and . - Calculate [H3O+] given pH: - Compare acid/base strengths: Use pKa and pKb values from tables.
Summary Table: Key Equations
Equation | Description |
|---|---|
Acid dissociation constant | |
Base dissociation constant | |
Ion-product constant for water | |
pH calculation | |
pOH calculation | |
pKa calculation | |
pKb calculation | |
Relationship between pKa and pKb |
Conclusion
Acid-base equilibria are central to understanding chemical reactions in aqueous solutions. Mastery of definitions, equilibrium constants, and calculations is essential for predicting reaction outcomes and understanding solution chemistry. Additional info: Academic context was added to clarify definitions, calculations, and relationships between acid/base strengths, as well as to provide self-contained explanations for exam preparation.
