 Back
BackAcid-Base Equilibria: Concepts, Calculations, and Molecular Structure
Study Guide - Smart Notes

Acids, Bases, and Conjugate Acid-Base Pairs
Brønsted-Lowry and Arrhenius Theories
The concept of acids and bases is fundamental in chemistry, with two primary theories describing their behavior: Arrhenius and Brønsted-Lowry. The Arrhenius theory defines acids as substances that increase the concentration of H+ ions in aqueous solution, and bases as substances that increase the concentration of OH– ions. The Brønsted-Lowry theory expands this definition, describing acids as proton donors and bases as proton acceptors.
Acid: Proton donor
Base: Proton acceptor
Conjugate acid-base pair: Two species that differ by a single proton (H+)
Example: Acetic acid and water reaction:
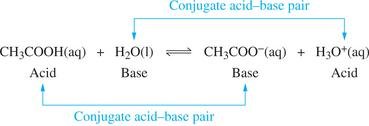
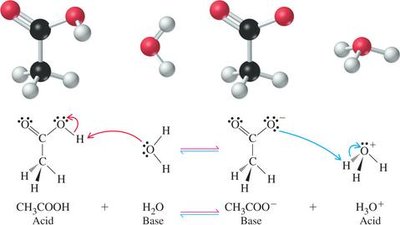
Example: Ammonia and water reaction:
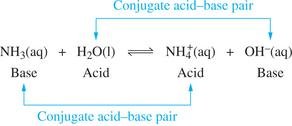
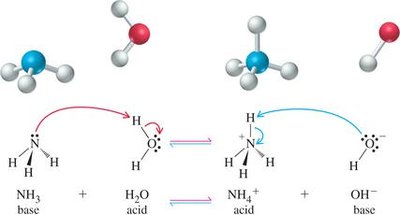
Key Point: Acids contain at least one ionizable hydrogen atom, while bases have an atom with a lone pair of electrons that can accept a proton.
Self-Ionization of Water and the pH Scale
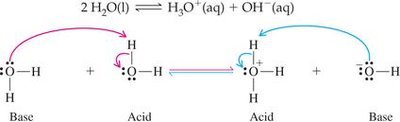
Self-Ionization of Water
Water can ionize itself, producing hydronium (H3O+) and hydroxide (OH–) ions. This equilibrium is crucial for understanding acid-base chemistry.
The ion product constant for water () at 25°C is:
In pure water at 25°C:
pH and pOH
The pH scale quantifies the acidity or basicity of a solution. It is defined as the negative logarithm of the hydronium ion concentration:
at 25°C

Example: Calculating pH and pOH from measured values.

Ionization of Acids and Bases in Water
Ionization Constants
Acids and bases ionize in water to varying degrees, characterized by their ionization constants:
Acid dissociation constant ():
Base dissociation constant ():
pK values: ,
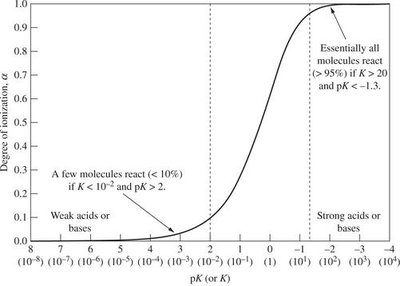
Degree of Ionization
The degree of ionization () measures the fraction of acid or base molecules that ionize:
for acids
for bases
Percent ionization:


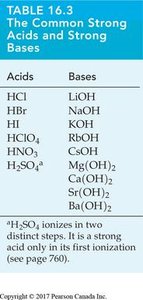
Strong Acids and Strong Bases
Properties and Examples
Strong acids and bases ionize completely in water, resulting in high concentrations of H3O+ or OH– ions. Their ionization constants are much greater than 1.
Strong acids: HCl, HBr, HI, HClO4, HNO3, H2SO4 (first ionization)
Strong bases: LiOH, NaOH, KOH, RbOH, CsOH, Ca(OH)2, Sr(OH)2, Ba(OH)2
Example: Calculating ion concentrations in strong acid/base solutions.


Weak Acids and Weak Bases
Properties and Calculations
Weak acids and bases only partially ionize in water. Their ionization constants are much less than 1, and equilibrium calculations are required to determine their concentrations.
Weak acids: Acetic acid, formic acid, etc.
Weak bases: Ammonia, pyridine, etc.



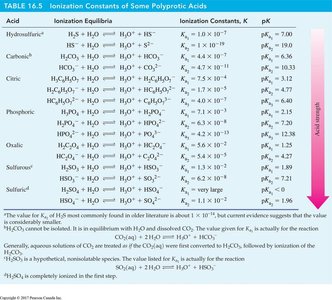
Polyprotic Acids
Definition and Ionization Steps
Polyprotic acids can donate more than one proton, ionizing in multiple steps, each with its own ionization constant. The first ionization is usually the strongest.
Example: Phosphoric acid () is triprotic.
Example: Sulfuric acid () is diprotic.



Ions as Acids and Bases
Conjugate Acid-Base Relationships
Ions can act as acids or bases depending on their ability to donate or accept protons. The relationship between the ionization constants of conjugate acid-base pairs is given by:
For a weak acid, its conjugate base is also weak. For a strong acid, its conjugate base is extremely weak.

Molecular Structure and Acid-Base Behavior
Binary Acids and Oxoacids
The strength of an acid depends on its molecular structure. For binary acids (HX), acid strength increases with bond polarity (across a period) and bond length (down a group). For oxoacids, strength increases with the electronegativity of the central atom and the number of terminal oxygen atoms.
Example: HI > HBr > HCl > HF (acid strength increases down the group)
Example: HNO3 is stronger than HNO2 due to more terminal oxygens.
Lewis Acids and Bases
Lewis Theory
The Lewis theory defines acids as electron pair acceptors and bases as electron pair donors. This theory broadens the concept of acids and bases beyond proton transfer.
Lewis acid: Electron pair acceptor
Lewis base: Electron pair donor
Strength of Amines as Bases
Amines are organic bases whose strength depends on their molecular structure and the availability of the nitrogen lone pair for protonation.
Summary Tables and Examples
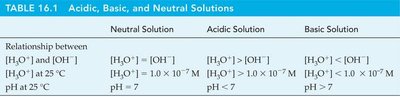
Acidic, Basic, and Neutral Solutions
The relationship between [H3O+] and [OH–] determines whether a solution is acidic, basic, or neutral.
Solution Type | [H3O+] vs [OH–] | pH |
|---|---|---|
Neutral | [H3O+] = [OH–] | pH = 7 |
Acidic | [H3O+] > [OH–] | pH < 7 |
Basic | [H3O+] < [OH–] | pH > 7 |

Relative Strengths of Acids and Bases
Acids and bases can be ranked by their ionization constants and pK values. Strong acids have high and low , while strong bases have high and low .
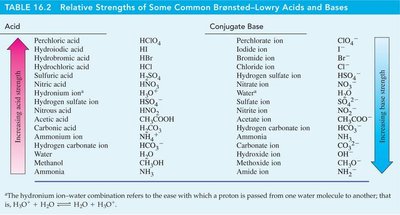

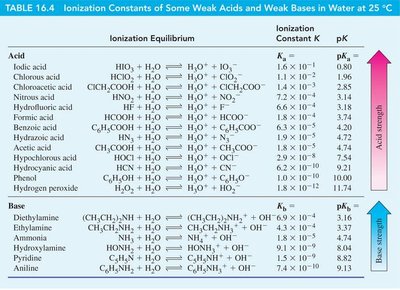
Worked Examples
Numerous worked examples illustrate calculations for identifying acids and bases, calculating pH, percent ionization, and ion concentrations in various types of solutions.













Additional info:
These notes cover the essential concepts of acid-base equilibria, including definitions, molecular structure, calculations, and the relationships between acids, bases, and their conjugates. The included images and tables reinforce key points and provide visual context for molecular interactions and calculations.
