 Back
BackAcid-Base Equilibria: Definitions, Calculations, and Examples
Study Guide - Smart Notes

Acid-Base Equilibria
Definitions of Acids and Bases
Acids and bases are fundamental concepts in chemistry, with several definitions that describe their behavior in different contexts:
Arrhenius Definition: Acids produce H+ ions (or H3O+) in aqueous solution, while bases produce OH– ions.
Brønsted-Lowry Definition: Acids are proton donors; bases are proton acceptors.
Lewis Definition: Acids accept electron pairs; bases donate electron pairs.
Arrhenius Acids and Bases
Arrhenius acids and bases are defined by their ability to increase the concentration of H+ or OH– ions in water:
Strong Acids: Completely dissociate in water. Examples: HCl, HBr, HI, HNO3, H2SO4, HClO4.
Weak Acids: Partially dissociate; not on the strong acid list.
Strong Bases: Completely dissociate in water. Examples: LiOH, NaOH, KOH, Ca(OH)2, Sr(OH)2, Ba(OH)2.
Weak Bases: Partially dissociate; often increase OH– indirectly (e.g., NH3).
Hydronium Ion Formation
H+ ions are unstable in water and react to form hydronium ions (H3O+):
Hydronium Ion: The H3O+ ion formed when an acid reacts with water.
H+ and H3O+ are often used interchangeably.

Acid-Base Neutralization
Acid-base neutralization reactions produce water and a salt:
General Reaction: Acid + Base → Water + Salt
Example: HNO3 (aq) + KOH (aq) → H2O (l) + KNO3 (aq)
Salt: An ionic compound formed from the cations and anions remaining after water is produced.
Brønsted-Lowry Acids and Bases
Brønsted-Lowry theory focuses on proton transfer:
Acid: Proton donor (e.g., HCl, H2SO4, HC2H3O2).
Base: Proton acceptor (e.g., OH–, NH3).
Amphiprotic: Water can act as either an acid or a base depending on the reaction.
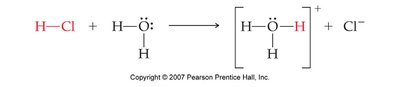
Conjugate Acid-Base Pairs
Acid-base reactions create pairs that differ by one proton:
Conjugate Acid: Formed when a base gains a proton.
Conjugate Base: Formed when an acid loses a proton.
Example: NH3 (aq) + H2O (l) ⇌ NH4+ (aq) + OH– (aq)
Autoionization of Water and Ion Product (Kw)
Water undergoes autoionization, producing H+ and OH– ions:
Reaction: H2O (l) ⇌ H+ (aq) + OH– (aq)
Ion Product: at 25°C
Kw: The equilibrium constant for water's autoionization.
Calculating [H+] and [OH–]
Knowing one ion concentration allows calculation of the other:
Formula:
Example: If [H+] = 1.8 × 10–5 M, then [OH–] = M
Equilibrium in Pure Water
In pure water, [H+] = [OH–], and the solution is neutral:
Calculation: , so M
Neutral Solution: pH = 7
pH Scale and Calculations
The pH scale is a convenient way to express acidity and basicity:
Definition:
Neutral: pH = 7
Acidic: pH < 7
Basic: pH > 7
Significant Figures: Only digits to the right of the decimal in pH are significant.
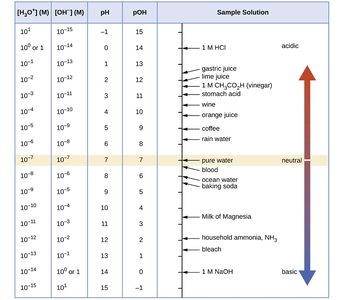
pOH and pKw
pOH is analogous to pH for hydroxide ions, and pKw relates both:
Definition:
pKw:
Relationship:
Example: If pH = 10.0, pOH = 4.0
Strong Acids and Bases: pH Calculations
For strong acids and bases, the ion concentration equals the solute concentration:
Example: 0.0142 M HBr → [H+] = 0.0142 M, pH =
Example: 1.5 M Sr(OH)2 → [OH–] = 3.0 M (since 2 OH– per formula unit)
Weak Acids and Bases: Equilibrium Calculations
Weak acids and bases do not fully dissociate; equilibrium expressions are used:
Acid Dissociation:
Ka (acid dissociation constant):
Base Dissociation:
Kb (base dissociation constant):
Polyprotic Acids
Polyprotic acids can donate more than one proton, each with its own dissociation constant:
Example: H2SO4 and H3PO4 have multiple dissociation steps.
Ka values: Each step has a unique Ka, decreasing with each proton removed.
Summary Table: Strong Acids and Bases
Strong Acids | Strong Bases |
|---|---|
HCl | LiOH |
HBr | NaOH |
HI | KOH |
HNO3 | Ca(OH)2 |
H2SO4 | Sr(OH)2 |
HClO4 | Ba(OH)2 |
Summary Table: pH, pOH, and Ion Concentrations
[H3O+] (M) | [OH–] (M) | pH | pOH | Sample Solution |
|---|---|---|---|---|
10–1 | 10–13 | 1 | 13 | 1 M HCl |
10–7 | 10–7 | 7 | 7 | Pure water |
10–14 | 10–1 | 14 | 0 | 1 M NaOH |
Additional info: See image_3 for more sample solutions and their pH values. | ||||
