 Back
BackAcid-Base Properties of Salt Solutions
Study Guide - Smart Notes

Acid-Base Properties of Salt Solutions
Introduction to Salt Solutions
Salt solutions can exhibit acidic, basic, or neutral properties depending on the nature of their constituent ions. Understanding the acid-base behavior of salts is essential for predicting the pH of their aqueous solutions. The process by which ions react with water to produce H+ or OH- is known as hydrolysis.
Hydrolysis: The reaction of ions with water to form H+ or OH-.
To determine the acid-base nature of a salt, analyze the cation and anion separately.
The cation can be acidic or neutral.
The anion can be acidic, basic, or neutral.

Acid-Base Behavior of Anions
Anions are the negatively charged ions in salts. Their acid-base behavior depends on their origin:
Anions of strong acids (e.g., Cl- from HCl) are neutral because they do not react with water to form OH-.
Anions of weak acids (e.g., CH3COO- from acetic acid) are basic because they can react with water to produce OH-.
Amphoteric anions can act as either acids or bases, depending on the conditions.

Acid-Base Behavior of Cations
Cations are the positively charged ions in salts. Their acid-base behavior is determined by their group and charge:
Group I and Group II metal cations (e.g., Na+, Ca2+, Sr2+, Ba2+) are neutral in solution.
Transition metal cations and polyatomic cations (e.g., NH4+) are often acidic due to their ability to hydrolyze water and release H+.

Acidity of Transition Metal Cations
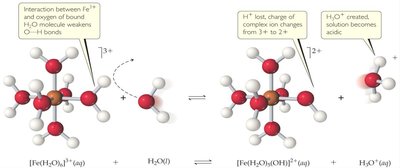
Transition metal cations, especially those with high charge, tend to be acidic in aqueous solution. This is due to their strong interaction with water molecules, which can lead to the release of H+ ions.
Highly charged cations (e.g., Fe3+, Al3+) polarize water molecules, weakening O–H bonds and facilitating proton release.
The resulting solution becomes acidic.
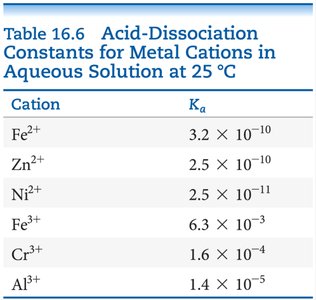
Acid-Dissociation Constants for Metal Cations
The acid-dissociation constant (Ka) quantifies the extent to which a metal cation can donate a proton to water. Higher Ka values indicate greater acidity.
Cation | Ka |
|---|---|
Fe2+ | 3.2 × 10-10 |
Zn2+ | 2.5 × 10-10 |
Ni2+ | 2.5 × 10-11 |
Fe3+ | 6.3 × 10-3 |
Cr3+ | 1.6 × 10-4 |
Al3+ | 1.4 × 10-5 |
Predicting the Acid-Base Nature of Salt Solutions
The acid-base properties of a salt solution depend on the combination of its cation and anion:
Group I/II metal cation + anion of a strong acid: Neutral solution
Group I/II metal cation + anion of a weak acid: Basic solution (like the anion)
Transition/post-transition metal cation or polyatomic cation + anion of a strong acid: Acidic solution (like the cation)

Examples and Applications
Consider the following salts and predict the nature of their aqueous solutions:
Ba(CH3COO)2: Ba2+ (Group II, neutral) + CH3COO- (anion of weak acid, basic) → Basic solution
NH4Cl: NH4+ (acidic) + Cl- (neutral) → Acidic solution
CH3NH3Br: CH3NH3+ (acidic) + Br- (neutral) → Acidic solution
KNO3: K+ (neutral) + NO3- (neutral) → Neutral solution
Al(ClO4)3: Al3+ (acidic) + ClO4- (neutral) → Acidic solution
Example: For NaH2PO4, H2PO4- is amphoteric, meaning it can act as both an acid and a base, depending on the conditions.
Aqueous Equilibrium Constants
The dissociation constants for acids and bases at 25°C are essential for quantifying their strength and predicting equilibrium positions in solution.

Summary Table: Salt Solution Properties
Salt Type | Cation | Anion | Resulting Solution |
|---|---|---|---|
NaCl | Na+ (neutral) | Cl- (neutral) | Neutral |
NH4Cl | NH4+ (acidic) | Cl- (neutral) | Acidic |
Ba(CH3COO)2 | Ba2+ (neutral) | CH3COO- (basic) | Basic |
Al(NO3)3 | Al3+ (acidic) | NO3- (neutral) | Acidic |
Key Equations
Hydrolysis of anion:
Hydrolysis of cation:
Acid-dissociation constant:
Additional info: Amphoteric ions, such as H2PO4-, can act as either acids or bases, depending on the pH and other ions present in solution.
