 Back
BackAcid-Base Properties of Salt Solutions, Molecular Structure and Acid Strength, and the Common Ion Effect
Study Guide - Smart Notes

Acid-Base Properties of Salt Solutions
How Salts Influence Solution Acidity and Basicity
Salt solutions can be acidic, basic, or neutral depending on the nature of their constituent ions. The cation and anion of a salt may react with water, affecting the pH of the solution.
Acidic Salt Solutions: Occur when the cation reacts with water to produce H3O+ ions, and the anion is inert (conjugate base of a strong acid).
Basic Salt Solutions: Occur when the anion reacts with water to produce OH- ions, and the cation is inert (conjugate acid of a strong base).
Neutral Salt Solutions: Occur when both ions are inert to water.
Example: NH4Cl is acidic because NH4+ reacts with water, while Cl- is inert.
Acidic Cations in Salts
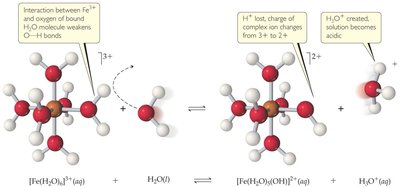
Cations that are conjugate acids of weak bases or small, highly charged metal ions (e.g., Fe3+, Al3+, Cr3+) can make salt solutions acidic.
Ammonium (NH4+): Reacts with water to produce NH3 and H3O+.
Highly Charged Metal Cations: Their strong positive charge polarizes water molecules, facilitating proton release and lowering pH.
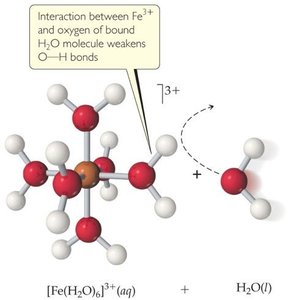

Acid-Dissociation Constants for Metal Cations
Metal cations differ in their ability to acidify solutions, as shown by their acid-dissociation constants (Ka).
Cation | Ka |
|---|---|
Fe2+ | 3.2 × 10-10 |
Zn2+ | 2.5 × 10-10 |
Ni2+ | 2.5 × 10-11 |
Fe3+ | 6.3 × 10-3 |
Cr3+ | 1.6 × 10-4 |
Al3+ | 1.4 × 10-5 |
Predicting Solution Acidity or Basicity
To predict whether a salt solution is acidic or basic, consider the reactivity of both cation and anion with water:
Acidic: NH4NO3, NaHSO4, Al(ClO4)3
Basic: Na2CO3, NaNO2
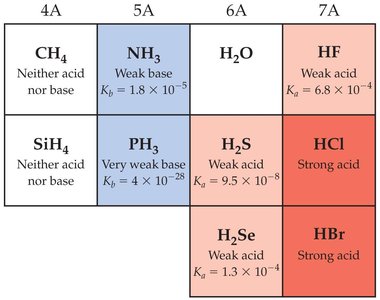
Molecular Structure and Acid Strength
Factors Affecting Acid Strength
The strength of an acid is determined by its molecular structure and the stability of its conjugate base.
Bond Polarity: Greater polarity in the H–A bond increases acidity.
Bond Strength: Weaker H–A bonds are easier to break, favoring acid dissociation.
Conjugate Base Stability: Stability is enhanced by electronegative atoms and resonance delocalization.
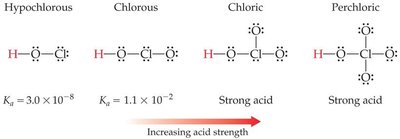
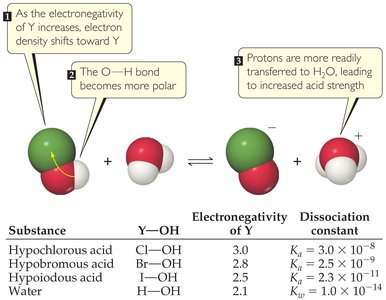
Acidity of Oxyacids
For oxyacids, acidity increases with the number of oxygen atoms bonded to the central atom and with the electronegativity of the central atom.
More Oxygens: Stabilizes the negative charge on the conjugate base.
Higher Electronegativity: Central atom pulls electron density, increasing bond polarity and acid strength.
Carboxylic Acids
Carboxylic acids are stronger acids than alcohols due to resonance stabilization of their conjugate base.
Resonance: Delocalizes negative charge, increasing stability and acidity.

The Common Ion Effect
Definition and Impact on Equilibria
The common ion effect occurs when a solution contains two substances that share a common ion. The presence of the common ion suppresses the ionization of the weak electrolyte, shifting equilibrium and affecting pH or solubility.
Acid Example: Adding NaF to HF(aq) reduces HF ionization, increasing pH.
Solubility Example: Adding NaCl to PbCl2 reduces PbCl2 solubility.
Quantitative Example: Hypochlorous Acid and NaClO
Adding NaClO to a solution of HClO increases ClO- concentration, shifting equilibrium and decreasing [H3O+], thus increasing pH.
ICE Table: Used to calculate new equilibrium concentrations.
Le Châtelier’s Principle: Addition of common ion shifts equilibrium to reduce ionization.
Chromate-Dichromate Equilibrium Demo
The equilibrium between chromate and dichromate ions is influenced by the addition of acids or bases, demonstrating the common ion effect.
Adding HCl: Increases [H+], shifts equilibrium toward dichromate (orange color).
Adding NaOH: Increases [OH-], shifts equilibrium toward chromate (yellow color).

Summary of Key Concepts
Salts can make solutions acidic or basic depending on their ions.
Acid strength is determined by molecular structure, bond polarity, bond strength, and conjugate base stability.
The common ion effect suppresses ionization or solubility when a common ion is added.
Equilibrium calculations and Le Châtelier’s Principle are essential for predicting changes in solution chemistry.
