 Back
BackAcid-Base Titration and pH: Concepts, Calculations, and Applications
Study Guide - Smart Notes

Acid-Base Titration and pH
Section 1: Aqueous Solutions and the Concept of pH
The study of acids, bases, and their reactions in aqueous solutions is fundamental to understanding chemical equilibria and solution chemistry. The pH scale provides a quantitative measure of the acidity or basicity of a solution, based on the concentration of hydronium ions.
Hydronium Ions and Hydroxide Ions
Self-ionization of Water: Water molecules can transfer protons, producing hydronium (H3O+) and hydroxide (OH−) ions.
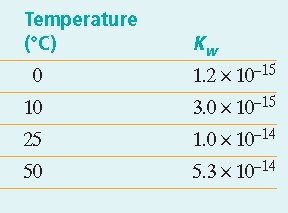
Ionization Constant of Water (Kw): The equilibrium constant for water ionization is given by: At 25°C,
Temperature Dependence: The value of increases with temperature, indicating greater ionization at higher temperatures.
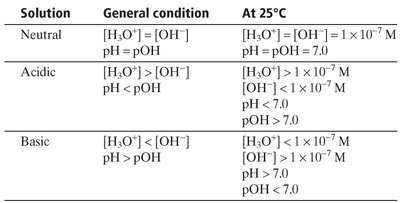
Neutral, Acidic, and Basic Solutions:
Neutral: M
Acidic:
Basic:
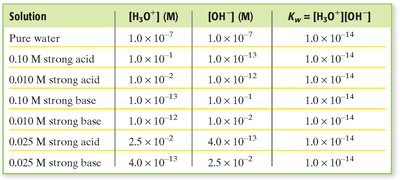
Strong Acids and Bases: These substances are considered completely ionized in aqueous solution. For example, a 0.010 M NaOH solution has M, and can be calculated using .
Sample Calculations
Given , calculate using :
Given , calculate similarly.
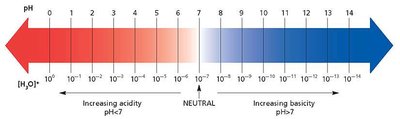
The pH Scale
The pH scale is a logarithmic scale used to specify the acidity or basicity of an aqueous solution.
Definition:
pOH:
Relationship: at 25°C
Neutral Solution: M,
Significant Figures: The number of decimal places in pH equals the number of significant figures in .
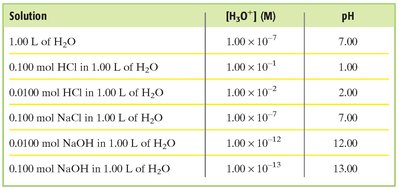
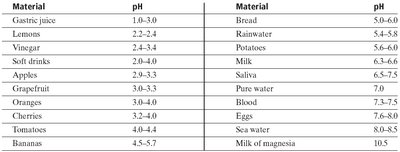
Common Materials and Their pH: Everyday substances have characteristic pH values, which can be referenced for practical understanding.
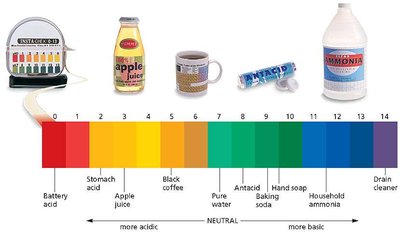
Calculating pH and [H3O+]
To find pH from :
To find from pH:

Relationship of [H3O+], [OH−], and pH
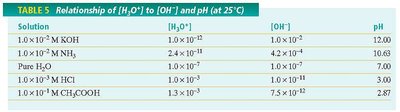

Section 2: Determining pH and Titrations
Indicators and pH Meters
Indicators: Compounds that change color depending on the pH of the solution. Each indicator has a specific transition interval (pH range over which it changes color).
pH Meters: Electronic devices that measure the voltage difference between electrodes to determine pH precisely.
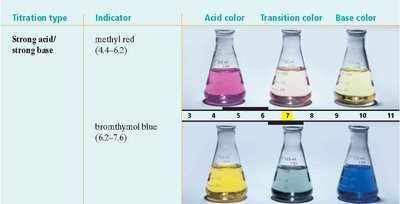
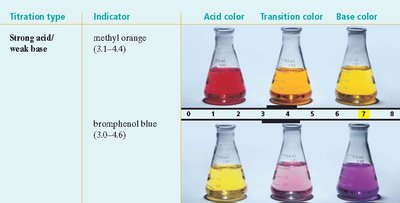
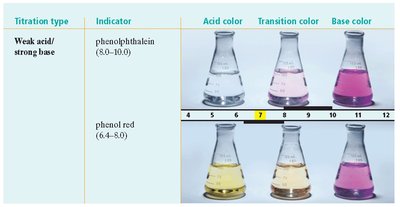
Titration and Equivalence Point
Titration: The process of adding a solution of known concentration (titrant) to a solution of unknown concentration until the reaction reaches completion (neutralization).
Equivalence Point: The point at which chemically equivalent amounts of acid and base have reacted.
End Point: The point at which the indicator changes color, signaling the completion of the titration.
Indicator Selection: The choice of indicator depends on the expected pH at the equivalence point (e.g., methyl red for strong acid/strong base, phenolphthalein for weak acid/strong base).
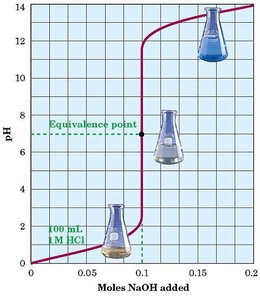
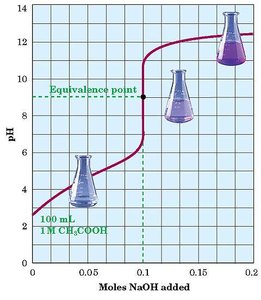
Molarity and Titration Calculations
Standard Solution: A solution of known concentration used in titration.
Primary Standard: A highly pure compound used to check the concentration of the standard solution.
Steps in Titration Calculations:
Write the balanced equation for the neutralization reaction.
Determine the moles of titrant used.
Calculate the moles of analyte (unknown solution) present.
Determine the molarity of the unknown solution using .
Acid and Base Strengths
Strong Acids/Bases: Completely ionize in solution (e.g., HCl, NaOH).
Weak Acids/Bases: Partially ionize; equilibrium exists between ionized and unionized forms.
Acid Dissociation Constant (Ka): Quantifies the strength of an acid in solution.
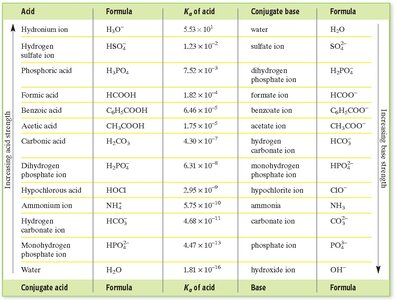
Additional info: The notes above integrate all major concepts, calculations, and tables relevant to acid-base titration and pH, as well as the use of indicators and titration curves. All images included are directly referenced and reinforce the adjacent explanations.
