 Back
BackAcid–Base Equilibria and Acid–Base Strength in Aqueous Solutions
Study Guide - Smart Notes

Acid–Base Equilibria
Introduction to Acid–Base Chemistry
Acid–base chemistry is fundamental to understanding chemical equilibria in aqueous solutions. Acids and bases can be defined in several ways, with the Arrhenius and Brønsted–Lowry definitions being the most common in general chemistry.
Arrhenius Acids: Substances that increase the concentration of H+ ions in water.
Arrhenius Bases: Substances that increase the concentration of OH− ions in water.
Brønsted–Lowry Acids: Proton (H+) donors.
Brønsted–Lowry Bases: Proton (H+) acceptors, typically possessing a lone pair of electrons.
Some molecules can act as both acids and bases (amphiprotic), such as water.
Acid–Base Reactions and Conjugate Pairs
In Brønsted–Lowry acid–base reactions, a proton is transferred from the acid to the base. Each acid–base pair is related by the gain or loss of a proton, forming conjugate acid–base pairs.
Conjugate Acid: The species formed when a base gains a proton.
Conjugate Base: The species formed when an acid loses a proton.
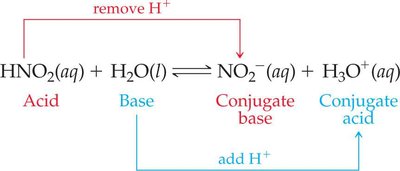
Example: In the reaction HNO2 + H2O ↔ NO2− + H3O+, HNO2 is the acid, H2O is the base, NO2− is the conjugate base, and H3O+ is the conjugate acid.
Acid and Base Strength
Strong and Weak Acids/Bases
The strength of an acid or base depends on its degree of ionization in water.
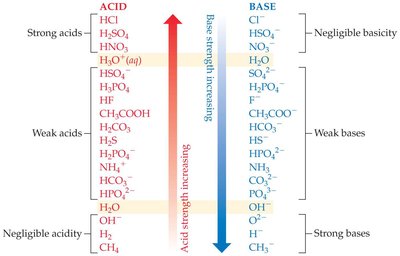
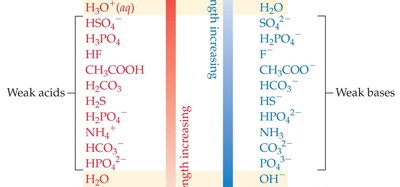
Strong Acids: Completely dissociate in water, producing a stoichiometric amount of H3O+. Their conjugate bases are very weak.
Weak Acids: Only partially dissociate in water, establishing an equilibrium with their conjugate bases.
Strong Bases: Completely dissociate in water to give OH− ions.
Weak Bases: Partially accept protons, establishing equilibrium with their conjugate acids.
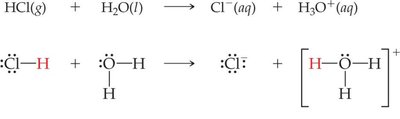
Example: HCl is a strong acid; CH3COOH (acetic acid) is a weak acid.
Equilibrium and Acid–Base Strength
Acid–base reactions are equilibria. The equilibrium favors the transfer of a proton from the stronger acid to the stronger base. For strong acids and bases, the equilibrium lies far to the right (products), while for weak acids and bases, the equilibrium is more balanced.


Autoionization of Water and the pH Scale
Autoionization of Water
Water is amphoteric and can undergo autoionization, producing hydronium and hydroxide ions:
The equilibrium constant for this process is called the ion product constant for water, .
at 25°C
The pH and pOH Scales
The pH scale is a logarithmic measure of hydronium ion concentration:
at 25°C
![pH, pOH, [H+], and [OH-] scale with common substances](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/3e4c4b9f_image_13.png)
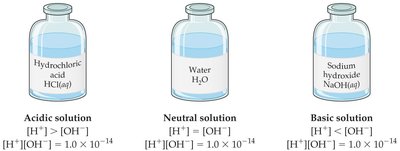
Neutral solution: [H+] = [OH−] = M, pH = 7.0
Acidic solution: [H+] > [OH−], pH < 7
Basic solution: [H+] < [OH−], pH > 7
Measuring pH
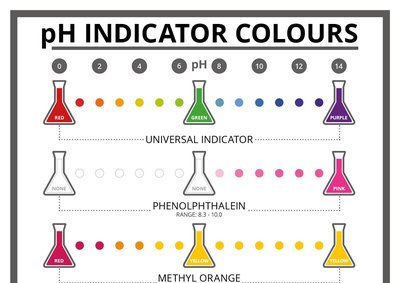
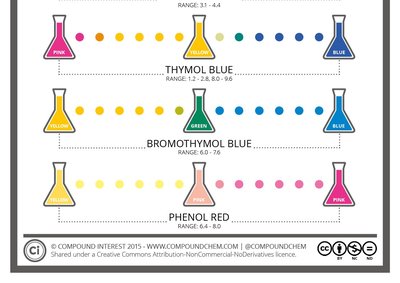
pH can be measured using indicators or electronic pH meters. Indicators change color depending on the pH of the solution, while pH meters provide a precise digital reading.


Calculating pH of Strong and Weak Acid/Base Solutions
Strong Acids and Bases
Strong acids and bases are completely dissociated in water. For monoprotic strong acids, [H3O+] = [acid] initial. For strong bases, [OH−] = [base] initial (or 2 × [base] for M(OH)2).


Weak Acids and Bases: Dissociation Constants
Weak acids and bases only partially dissociate, and their strength is quantified by the acid dissociation constant () or base dissociation constant ().
For a weak acid HA:
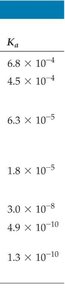
The larger the , the stronger the acid.
Calculating pH from
To calculate the pH of a weak acid solution:
Write the balanced equation for dissociation.
Set up an ICE (Initial, Change, Equilibrium) table.
Solve for [H3O+] using the expression, often making the approximation that x is small compared to the initial concentration.
Calculate pH from [H3O+].
Example: For 0.010 M niacin (), set up the equilibrium and solve for x, then pH = –log(x).
Calculating from pH
If the pH and initial concentration are known, can be calculated by determining [H3O+] from pH and using the equilibrium concentrations in the expression.
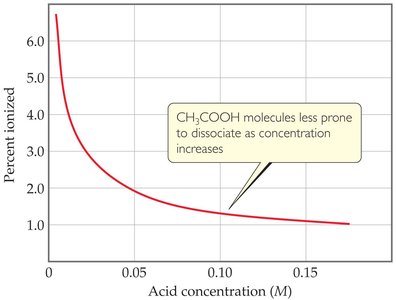
Percent Ionization
Percent ionization indicates the fraction of acid molecules that ionize in solution:
Percent ionization =
As the concentration of a weak acid decreases, percent ionization increases.
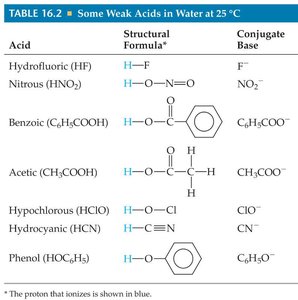
Summary Table: Some Weak Acids and Their Values
Acid | Structural Formula | Conjugate Base | |
|---|---|---|---|
Hydrofluoric (HF) | H–F | F− | 6.8 × 10−4 |
Nitrous (HNO2) | H–O–N=O | NO2− | 4.5 × 10−4 |
Benzoic (C6H5COOH) | See structure | C6H5COO− | 6.3 × 10−5 |
Acetic (CH3COOH) | See structure | CH3COO− | 1.8 × 10−5 |
Hypochlorous (HClO) | H–O–Cl | ClO− | 3.0 × 10−8 |
Hydrocyanic (HCN) | H–C≡N | CN− | 4.9 × 10−10 |
Phenol (HOC6H5) | See structure | HOC6H5O− | 1.3 × 10−10 |
