 Back
BackAcids and Bases: Concepts, Calculations, and Molecular Structure
Study Guide - Smart Notes

Acids and Bases: Fundamental Concepts
Introduction to Acids and Bases
Acids and bases are central to many chemical reactions and are commonly encountered in both laboratory and everyday contexts. The Brønsted–Lowry theory provides a modern framework for understanding acid–base behavior, defining acids as proton donors and bases as proton acceptors. Many substances, including those found in citrus fruits, owe their properties to the presence of acids such as citric acid and ascorbic acid (vitamin C).

Brønsted–Lowry Theory and Conjugate Acid–Base Pairs
Definition and Examples
The Brønsted–Lowry theory defines an acid as a substance that donates a proton (H+) and a base as a substance that accepts a proton. Acid–base reactions are reversible and involve the transfer of protons between reactants. Each acid has a corresponding conjugate base (formed by loss of a proton), and each base has a conjugate acid (formed by gain of a proton). These pairs differ by a single proton.
Example: The ionization of acetic acid in water: - CH3COOH: acid (proton donor) - H2O: base (proton acceptor) - CH3COO-: conjugate base - H3O+: conjugate acid
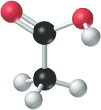
Amphiprotic Substances
Some substances, such as water, can act as either an acid or a base depending on the reaction context. These are called amphiprotic substances.
Self-Ionization of Water and the pH Scale
Self-Ionization (Autoionization) of Water
Water can ionize to a small extent, producing hydronium and hydroxide ions:
The equilibrium constant for this reaction is called the ion product of water ():
at 25°C
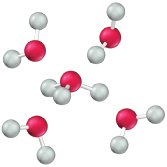
pH and pOH
The pH of a solution is a measure of its acidity, defined as:
Similarly, pOH is defined as:
At 25°C, .
Ionization of Acids and Bases in Water
Strong vs. Weak Acids and Bases
Strong acids/bases ionize completely in water (e.g., HCl, NaOH).
Weak acids/bases ionize only partially (e.g., CH3COOH, NH3).
The acid ionization constant () and base ionization constant () quantify the extent of ionization:
The degree of ionization () and percent ionization are used to describe how much of an acid or base ionizes in solution.
Experimental Comparison of Strong and Weak Acids
Strong acids produce higher concentrations of hydronium ions than weak acids at the same molarity, as shown by pH measurements and acid–base indicators.

Structure and Strength of Acids and Bases
Carboxylic Acids and Amines
Many weak acids, such as acetic acid and lactic acid, contain the carboxyl group (–COOH). Weak bases often contain nitrogen atoms, as in amines.
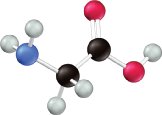
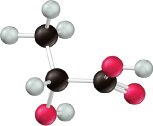
Organic Acids and Substituent Effects
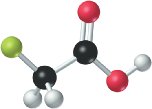
The presence of electronegative atoms (e.g., Cl, F) near the acidic hydrogen increases acid strength by stabilizing the conjugate base. For example, fluoroacetic acid is stronger than acetic acid due to the electron-withdrawing effect of fluorine.
Polyprotic Acids
Stepwise Ionization
Polyprotic acids, such as phosphoric acid (H3PO4), can donate more than one proton in a stepwise manner. Each ionization step has its own value, with the first ionization being the largest.


pH of Salt Solutions and Hydrolysis
Hydrolysis of Ions
Salts formed from weak acids or bases can hydrolyze in water, affecting the pH. For example, sodium acetate (from acetic acid) makes the solution basic, while ammonium chloride (from ammonia) makes it acidic.

Lewis Acids and Bases
Electron-Pair Donors and Acceptors
The Lewis definition broadens the concept of acids and bases. A Lewis acid is an electron-pair acceptor, and a Lewis base is an electron-pair donor. This theory explains acid–base reactions that do not involve protons, such as the formation of complex ions.

Summary Table: Key Relationships
Type | Definition | Example |
|---|---|---|
Brønsted–Lowry Acid | Proton donor | HCl, CH3COOH |
Brønsted–Lowry Base | Proton acceptor | NH3, H2O |
Lewis Acid | Electron-pair acceptor | BF3, Al3+ |
Lewis Base | Electron-pair donor | NH3, H2O |
Strong Acid | Completely ionizes in water | HCl, HNO3 |
Weak Acid | Partially ionizes in water | CH3COOH |
Key Equations
(at 25°C)
(for conjugate acid–base pairs)
Additional info:
Many calculations involving weak acids and bases use simplifying assumptions, but these must be checked for validity (e.g., the 5% rule).
The structure and stability of conjugate bases/anions play a major role in determining acid strength, especially in organic and polyprotic acids.
