 Back
BackAcids and Bases: Concepts, Strength, and Titration
Study Guide - Smart Notes

Acids and Bases
Introduction to Acids and Bases
Acids and bases are fundamental chemical species that participate in a wide range of chemical reactions. Their behavior can be described by several theoretical models, each providing a different perspective on what constitutes an acid or a base.
Acid: A substance that can donate a proton (H+) or increase the concentration of H+ ions in solution.
Base: A substance that can accept a proton or increase the concentration of OH- ions in solution.
Example: The reaction between hydrochloric acid (HCl) and ammonia (NH3) forms ammonium chloride (NH4Cl), demonstrating acid-base behavior.

Acid-Base Theories
Arrhenius Concept
The Arrhenius definition is one of the earliest models for acids and bases, focusing on their behavior in aqueous solutions.
Arrhenius Acid: Increases the concentration of H+ (or H3O+) ions in water. Example: HCl.
Arrhenius Base: Increases the concentration of OH- ions in water. Example: NaOH, Ca(OH)2.
Limitation: Some substances, like NH3, increase pH without directly donating OH- ions.
Brønsted-Lowry Concept
The Brønsted-Lowry theory broadens the definition of acids and bases to include proton transfer reactions.
Brønsted-Lowry Acid: Proton (H+) donor.
Brønsted-Lowry Base: Proton (H+) acceptor.
Key Point: Acid-base behavior depends on the presence of a suitable reaction partner; no substance is inherently an acid or base.
Example: In the reaction HCl + NH3 → NH4+ + Cl-, HCl donates a proton (acid), NH3 accepts it (base).
Conjugate Acid-Base Pairs
Definition and Examples
Conjugate acid-base pairs are species that differ by one proton. When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid.
Conjugate Acid: The species formed when a base gains a proton.
Conjugate Base: The species formed when an acid loses a proton.
Example: NH3 (base) + H+ → NH4+ (conjugate acid).
The pH Scale
Definition and Interpretation
The pH scale is a logarithmic scale used to quantify the acidity or basicity of an aqueous solution. It is defined as:
pH = –log10[H+]
Acidic solutions: pH < 7
Neutral solutions: pH = 7
Basic solutions: pH > 7

Autoprotolysis of Water
Self-Ionization and Amphiprotic Nature
Water can act as both an acid and a base, a property known as amphiprotic behavior. In pure water, a small fraction of molecules undergo autoprotolysis (self-ionization):
Ion product of water: at 25°C
pH + pOH = 14 at 25°C
Water as an ampholyte: Can both donate and accept protons.

Acid and Base Strength
Quantifying Strength: Ka, Kb, pKa, pKb
The strength of an acid or base is measured by its tendency to donate or accept protons, quantified by equilibrium constants:
Acid dissociation constant (Ka):
Base dissociation constant (Kb):
pKa = –log Ka
pKb = –log Kb
Relationship: and
Interpretation: Lower pKa means a stronger acid; lower pKb means a stronger base.

Relative Strengths of Acids and Bases
Acids and bases can be classified as strong or weak depending on their degree of ionization in water.
Strong acids: Completely ionize in water (e.g., HCl, HNO3).
Weak acids: Partially ionize in water (e.g., acetic acid).
Strong bases: Completely dissociate in water (e.g., NaOH, KOH).
Weak bases: Partially accept protons (e.g., NH3).



Polyprotic Acids
Definition and Behavior
Polyprotic acids are acids that can donate more than one proton per molecule. Each deprotonation step has its own equilibrium constant and pKa value, which increases with each successive proton loss.
Examples: Sulfuric acid (H2SO4), phosphoric acid (H3PO4).
Each step:
pKa values: Increase with each deprotonation, indicating decreasing acid strength.
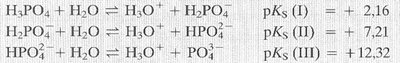
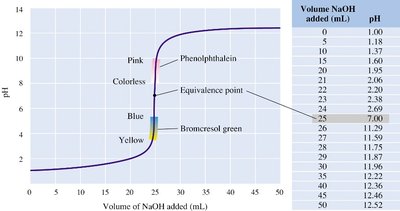
Titrations
Principles and Curves
Titration is an analytical technique used to determine the concentration of an unknown acid or base by reacting it with a standard solution of known concentration. The progress of the titration is monitored by measuring the pH as a function of the volume of titrant added.
Titration curve: A plot of pH versus volume of titrant added.
Equivalence point: The point at which stoichiometrically equivalent amounts of acid and base have reacted.
Indicators: Substances that change color at specific pH values to signal the endpoint of the titration.
Summary Table: Acid and Base Strengths
The following tables summarize the relative strengths and dissociation constants of common acids and bases:
Strong Acids | Strong Bases |
|---|---|
HClO4, H2SO4, HI, HBr, HCl, HNO3 | LiOH, NaOH, KOH, Ca(OH)2, Sr(OH)2, Ba(OH)2 |
Acid | Base |
|---|---|
HClO4 | ClO4- |
H2SO4 | HSO4- |
HI | I- |
HBr | Br- |
HCl | Cl- |
HNO3 | NO3- |
... | ... |
*Additional info: For a complete list, refer to the provided tables in the original material.*
