 Back
BackAcids and Bases: Definitions, Properties, and Calculations
Study Guide - Smart Notes

Acids and Bases
Definitions of Acids and Bases
Acids and bases are fundamental concepts in chemistry, with several definitions that help classify their behavior in different contexts. The three main definitions are Arrhenius, Brønsted-Lowry, and Lewis.
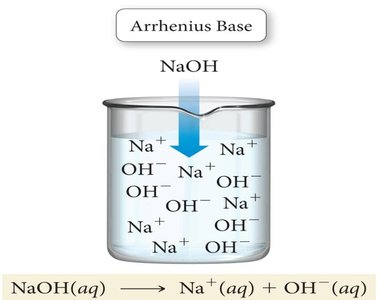
Arrhenius Acids and Bases: An Arrhenius acid is a substance that produces H+ ions in water, while an Arrhenius base produces OH− ions in water.
Example: Dissolving HCl in water produces H+ and Cl− ions. Dissolving NaOH produces Na+ and OH− ions.
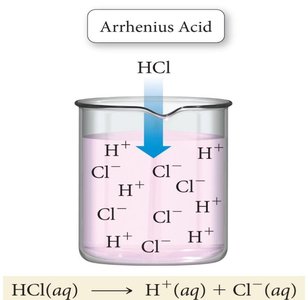
Hydrogen Ion and Hydronium Ion: In aqueous solution, H+ does not exist freely but forms H3O+ (hydronium ion). For simplicity, H+ and H3O+ are used interchangeably.
Neutralization Reaction:
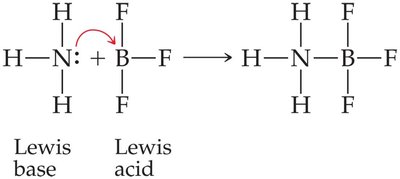
Lewis Acids and Bases: A Lewis acid is an electron pair acceptor, and a Lewis base is an electron pair donor.
Example: (NH3 is the Lewis base, BF3 is the Lewis acid).
Brønsted-Lowry Acids and Bases: A Brønsted-Lowry acid is a proton (H+) donor, and a Brønsted-Lowry base is a proton acceptor. Acid-base reactions always involve both an acid and a base.
Example:
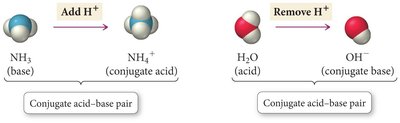
Conjugate Acid-Base Pairs: When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid.
Example: NH3 (base) becomes NH4+ (conjugate acid); H2O (acid) becomes OH− (conjugate base).
Additional info: Some substances can act as both acids and bases (amphoteric), and acid-base reactions can occur in non-aqueous solvents and the gas phase.
Water as a Solvent
Auto-Ionization and Ion-Product Constant
Water is amphoteric, meaning it can act as both an acid and a base. This property allows water to undergo auto-ionization, producing hydronium and hydroxide ions.
Auto-Ionization Reaction: or
Ion-Product Constant (Kw): at 25°C
In a neutral solution, M.
pH Scale and Measurements
pH, pOH, and Their Calculations
The pH scale is a logarithmic measure of the hydronium ion concentration in a solution. It is used to express the acidity or basicity of a solution.
pH Definition:
pOH Definition:
Relationship: at 25°C
Hydronium Ion Concentration:
Hydroxide Ion Concentration:
Example: If pH = 4.70, M. The solution is acidic.
Example: If M, , (basic solution).
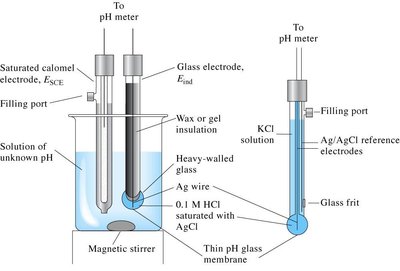
pH Measurement: pH is measured using a pH meter, which uses a glass membrane electrode sensitive to [H3O+].
Weak Acids (Monoprotic): Acid Dissociation Constant (Ka), % Ionization, pH Calculations
Strong vs. Weak Acids
Acids are classified as strong or weak based on their degree of ionization in water. Strong acids ionize completely, while weak acids ionize partially.
Strong Acids: Ionize 100% in water. Examples include HCl, HBr, HI, HNO3, HClO4, HClO3, and H2SO4 (first proton only).
pH Calculation for Strong Acids: ;
Example: 0.10 M HCl yields pH = 1.00.
Weak Acids and Acid Dissociation Constant (Ka)
Weak acids only partially ionize in water. The extent of ionization is quantified by the acid dissociation constant, Ka.
Ionization Reaction:
Equilibrium Expression:
pKa: ; smaller pKa means stronger acid.
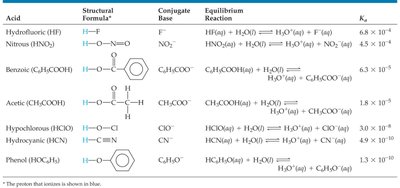
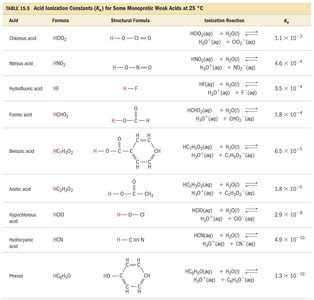
Degree of Ionization: The larger the Ka, the stronger the acid and the higher its % ionization.
% Ionization Formula:
Effect of Dilution on % Ionization
As the concentration of a weak acid decreases (dilution), the % ionization increases because the equilibrium shifts to produce more H3O+.
Calculating pH of a Weak Acid Solution
To calculate the pH of a weak acid solution, use the Ka expression and make appropriate assumptions about the degree of ionization.
Example: For 0.15 M acetic acid (Ka = 1.8 × 10−5):
Additional info: The assumption that x is negligible compared to the initial concentration is valid if % ionization is ≤ 5%. Otherwise, the quadratic equation must be used.
