 Back
BackAcids and Bases: Definitions, Properties, and Calculations
Study Guide - Smart Notes

Chapter 17: Acids and Bases
Definitions of Acids and Bases
Understanding acids and bases is fundamental in chemistry. Several definitions exist, each broadening the scope of what constitutes an acid or a base.
Arrhenius Acids and Bases
Arrhenius Acid: A substance that, when dissolved in water, increases the concentration of hydrogen ions (H+).
Arrhenius Base: A substance that, when dissolved in water, increases the concentration of hydroxide ions (OH−).
Example: HCl is an Arrhenius acid because it dissociates in water to produce H+ and Cl− ions. NaOH is an Arrhenius base because it dissociates to produce Na+ and OH− ions.
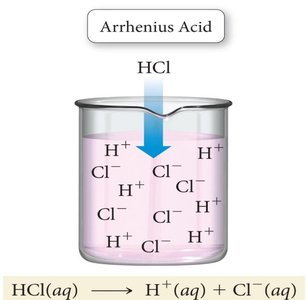
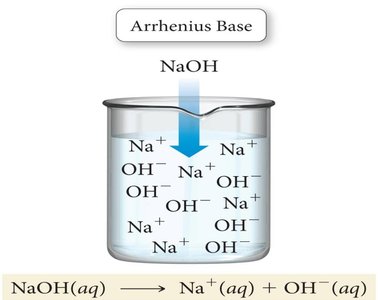
Hydrogen Ion and Hydronium Ion
In aqueous solution, the hydrogen ion (H+) does not exist freely but bonds to water to form the hydronium ion (H3O+).
For simplicity, H+(aq) and H3O+(aq) are used interchangeably.
Neutralization Reaction: H+(aq) + OH−(aq) → H2O(l)
Lewis Acids and Bases
Lewis Acid: An electron pair acceptor.
Lewis Base: An electron pair donor.
Many metal ions act as Lewis acids due to their empty orbitals.
Example: NH3 (Lewis base) donates a pair of electrons to BF3 (Lewis acid) to form an adduct.
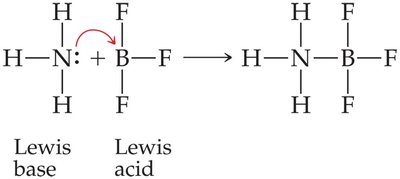
Brønsted-Lowry Acids and Bases
Brønsted-Lowry Acid: A proton (H+) donor.
Brønsted-Lowry Base: A proton (H+) acceptor.
Acid-base reactions always involve a pair: one species donates a proton, the other accepts it.
These reactions can occur in aqueous and non-aqueous solutions, and even in the gas phase.
Example: NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH−(aq)
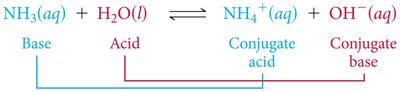
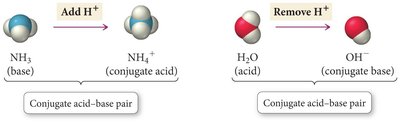
The Conjugate Acid-Base Concept
Every acid forms a conjugate base after donating a proton.
Every base forms a conjugate acid after accepting a proton.
Acid + Base ⇌ Conjugate Base + Conjugate Acid
Water as a Solvent
Auto-Ionization of Water
Water is amphoteric: it can act as both an acid and a base.
Water undergoes auto-ionization (self-ionization):
Or simply:
Ion-Product Constant of Water, Kw
The equilibrium constant for water is:
At 25°C,
In neutral water: M
pKw, pH, and pOH
pH:
pOH:
Relationship: at 25°C
As [H3O+] increases, pH decreases (more acidic).
pH Scale and Measurements
The pH scale is a logarithmic scale used to express the acidity or basicity of a solution.
pH normally ranges from 0 (very acidic) to 14 (very basic), but values outside this range are possible.
Each unit change in pH represents a tenfold change in [H+].
pOH is used similarly for hydroxide ion concentration.
Example: If [OH−] = 1.0 × 10−6 M, then pOH = 6.00.
pH + pOH = 14.00 at 25°C.
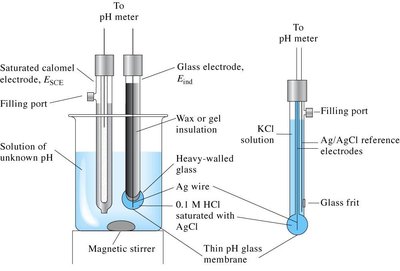
pH Measurements
pH is measured using a pH meter, which uses a glass electrode sensitive to [H3O+].
Less accurate methods include pH paper and indicators.



Weak Acids (Monoprotic): Acid Dissociation Constant (Ka), % Ionization, and pH Calculations
Strong Acids
Strong acids ionize completely in water; [H+] equals the acid's molarity.
Example: 0.10 M HCl yields [H3O+] = 0.10 M, so pH = 1.00.
Common strong acids: HCl, HBr, HI, HNO3, HClO4, HClO3, H2SO4 (first proton only).
Weak Acids
Weak acids ionize only partially in water.
General reaction: HA(aq) + H2O(l) ⇌ H3O+(aq) + A−(aq)
The acid dissociation constant (Ka) quantifies acid strength:
Larger Ka = stronger acid; smaller Ka = weaker acid.
pKa = −log Ka; smaller pKa = stronger acid.

% Ionization of a Weak Acid
% Ionization = ([H3O+]eq / [HA]i) × 100
As acid is diluted, % ionization increases.

Calculating pH of a Weak Acid Solution
Set up an ICE table (Initial, Change, Equilibrium) for the reaction.
Assume x (amount ionized) is small if % ionization ≤ 5%.
Solve for x using Ka expression; x = [H3O+].
Calculate pH: pH = −log [H3O+].
If % ionization > 5%, use the quadratic formula for accuracy.
Example: For 0.15 M acetic acid (Ka = 1.8 × 10−5):
% Ionization = (1.6 × 10−3 / 0.15) × 100 = 1.1%
pH = −log(1.6 × 10−3) = 2.80
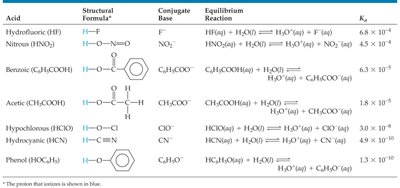
Summary Table: Acid Dissociation Constants for Selected Weak Acids
Acid | Formula | Structural Formula | Conjugate Base | Equilibrium Reaction | Ka |
|---|---|---|---|---|---|
Hydrofluoric acid | HF | H–F | F− | HF(aq) + H2O(l) ⇌ H3O+(aq) + F−(aq) | 6.8 × 10−4 |
Nitrous acid | HNO2 | H–O–N=O | NO2− | HNO2(aq) + H2O(l) ⇌ H3O+(aq) + NO2−(aq) | 4.5 × 10−4 |
Benzoic acid | C6H5COOH | Ph–COOH | C6H5COO− | C6H5COOH(aq) + H2O(l) ⇌ H3O+(aq) + C6H5COO−(aq) | 6.3 × 10−5 |
Acetic acid | CH3COOH | CH3–COOH | CH3COO− | CH3COOH(aq) + H2O(l) ⇌ H3O+(aq) + CH3COO−(aq) | 1.8 × 10−5 |
Hypochlorous acid | HClO | H–Cl–O | ClO− | HClO(aq) + H2O(l) ⇌ H3O+(aq) + ClO−(aq) | 3.0 × 10−8 |
Hydrocyanic acid | HCN | H–C≡N | CN− | HCN(aq) + H2O(l) ⇌ H3O+(aq) + CN−(aq) | 4.9 × 10−10 |
Phenol | C6H5OH | Ph–OH | C6H5O− | C6H5OH(aq) + H2O(l) ⇌ H3O+(aq) + C6H5O−(aq) | 1.3 × 10−10 |
