 Back
BackAcids and Bases: Properties, Definitions, and Reactions
Study Guide - Smart Notes

Acids and Bases
Introduction to Acids and Bases
Acids and bases are fundamental classes of compounds in chemistry, each with distinct properties and important roles in both laboratory and everyday contexts. Their behavior is central to many chemical reactions, including those in biological and industrial processes.
Properties of Acids
Sour Taste: Acids typically taste sour. For example, the sour taste of candies like Sour Patch Kids is due to citric and tartaric acids, which release H+ ions that interact with taste receptors.
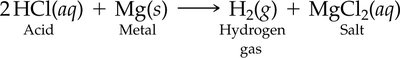
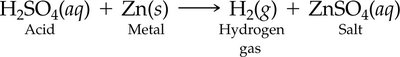
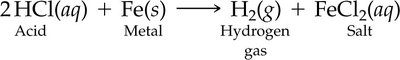
Reaction with Metals: Acids can dissolve many metals, producing hydrogen gas and a salt. However, some metals like gold are resistant to acid attack.
Litmus Test: Acids turn blue litmus paper red.


Examples of Common Acids
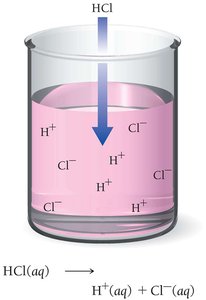
Hydrochloric Acid (HCl): Found in laboratories and stomach acid; used in cleaning metals and food processing.
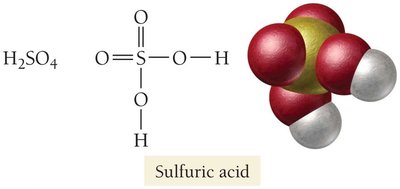
Sulfuric Acid (H2SO4): Widely produced for fertilizers, batteries, and industrial processes.
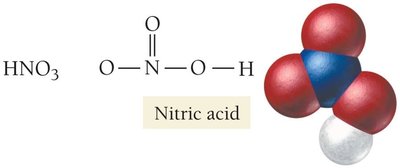
Nitric Acid (HNO3): Used in fertilizers and explosives.
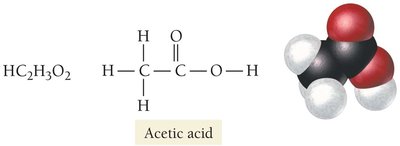
Acetic Acid (HC2H3O2): Main component of vinegar; a carboxylic acid.
Carboxylic Acids: Organic acids containing the –COOH group, found in many biological substances (e.g., citric acid in lemons, malic acid in apples).

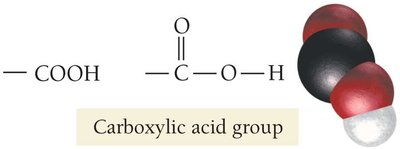
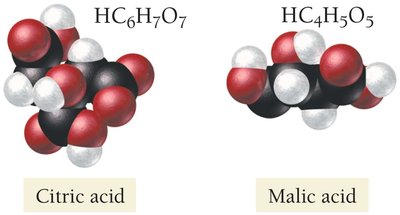
Properties of Bases
Bitter Taste: Bases taste bitter, which is a natural deterrent against consuming potentially toxic substances (e.g., alkaloids like coniine).
Slippery Feel: Bases feel slippery because they react with oils on the skin to form soap-like substances.
Litmus Test: Bases turn red litmus paper blue.

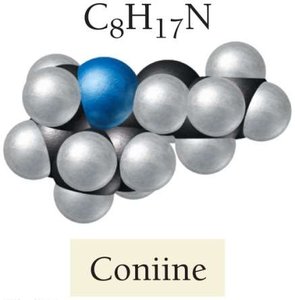
Examples of Common Bases
Sodium Hydroxide (NaOH): Used in drain cleaners and soap manufacturing.
Potassium Hydroxide (KOH): Used in similar applications as NaOH.
Sodium Bicarbonate (NaHCO3): Baking soda; used as an antacid to neutralize stomach acid.
Definitions of Acids and Bases
Arrhenius Definition
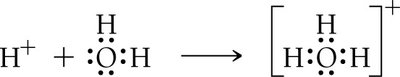
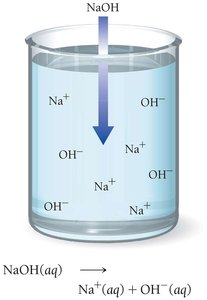
Acid: Produces H+ ions in aqueous solution.
Base: Produces OH− ions in aqueous solution.
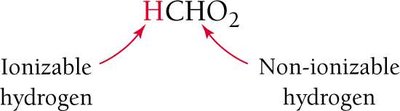
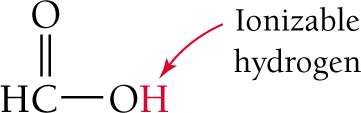
In the molecular formula for an acid, the ionizable hydrogen is often written first (e.g., HCHO2 for formic acid).
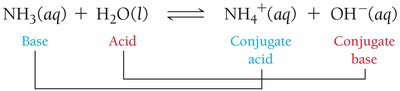
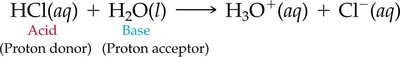
Brønsted–Lowry Definition
Acid: Proton (H+) donor.
Base: Proton (H+) acceptor.
This definition is broader and applies to more reactions, including those not in water. For example, ammonia (NH3) acts as a base by accepting a proton from water.
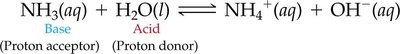
Amphoteric Substances and Conjugate Pairs
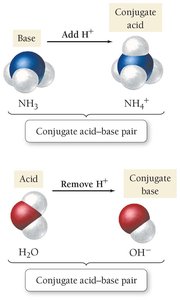
Amphoteric: Substances like water can act as either an acid or a base.
Conjugate Acid–Base Pair: Two substances related by the gain or loss of a proton (e.g., NH3 and NH4+).
Reactions of Acids and Bases
Neutralization Reactions
When an acid and a base react, they form water and a salt. The net ionic equation for many neutralization reactions is:
Reactions of acids with carbonates or bicarbonates produce water, carbon dioxide gas, and a salt (gas evolution reaction).

Acids Reacting with Metals and Metal Oxides
Acids react with many metals to produce hydrogen gas and a salt.
Some metals, such as gold, do not react with acids.
Acids also react with metal oxides to produce water and a dissolved salt.

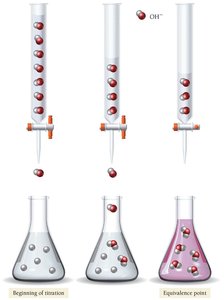
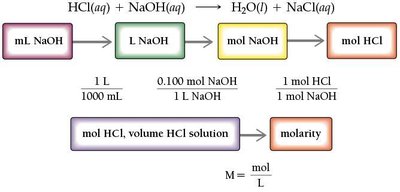
Acid–Base Titration
Titration is a laboratory technique used to determine the concentration of an unknown acid or base by reacting it with a solution of known concentration. The equivalence point is reached when the amount of acid equals the amount of base, as indicated by a color change from an indicator such as phenolphthalein.


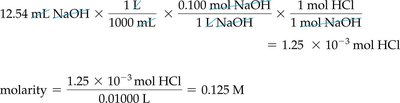
Strong and Weak Acids and Bases
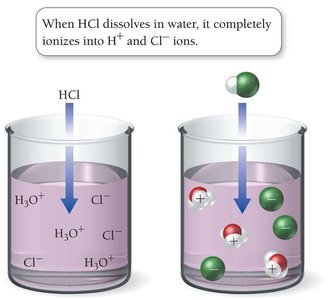
Strong Acids
Completely ionize in solution, producing a high concentration of H3O+ ions.
Examples: HCl, HBr, HI, HNO3, HClO4, H2SO4 (first proton only).


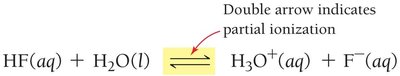
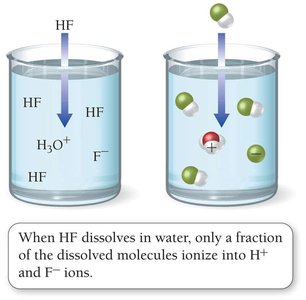
Weak Acids
Only partially ionize in solution; most molecules remain intact.
Examples: HF, acetic acid, formic acid, carbonic acid, phosphoric acid.

Strong Bases
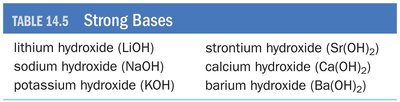
Completely dissociate in solution to produce OH− ions.
Examples: NaOH, KOH, Ba(OH)2, Sr(OH)2.
Weak Bases
Partially react with water to produce OH− ions; most molecules remain unreacted.
Examples: Ammonia (NH3), organic amines.
Water: Acid and Base in One
Self-Ionization of Water
Water is amphoteric and can act as both an acid and a base. In pure water, a small amount of self-ionization occurs:
At 25°C, , and the ion product constant is:
Acidic, Basic, and Neutral Solutions
Neutral:
Acidic: ,
Basic: ,
The pH and pOH Scales
Definition and Calculation
pH:
pOH:
At 25°C: pH + pOH = 14
pH < 7: acidic; pH > 7: basic; pH = 7: neutral
Buffers
Definition and Function
A buffer is a solution that resists changes in pH when small amounts of acid or base are added. Buffers contain significant amounts of both a weak acid and its conjugate base. They are essential in biological systems, such as human blood, to maintain a stable pH.
Chemistry and Health: Acid Rain and Antifreeze Poisoning
Acid Rain: Caused by sulfur and nitrogen oxides from fossil fuels, which form acids in the atmosphere and damage buildings and ecosystems.
Antifreeze Poisoning: Ethylene glycol is metabolized to glycolic acid, overwhelming the body's buffer system and leading to dangerous drops in blood pH.
