 Back
BackAcids and Bases: Properties, Definitions, and Calculations
Study Guide - Smart Notes

Acids and Bases
Introduction to Acids and Bases
Acids and bases are fundamental classes of compounds in chemistry, playing crucial roles in chemical reactions, biological systems, and industrial processes. Their properties, definitions, and behaviors are central to understanding chemical equilibrium and solution chemistry.
Properties and Examples of Acids
General Properties of Acids
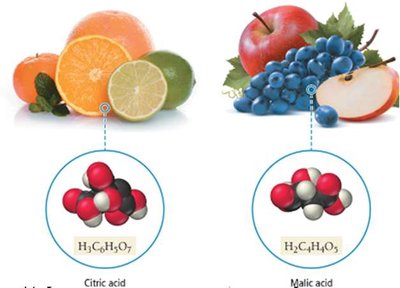
Sour taste (e.g., citric acid in lemons)
Ability to dissolve many metals (e.g., HCl reacts with Zn to produce H2 gas)
Ability to neutralize bases
Change blue litmus paper to red
Common Acids and Their Uses
Name | Formula | Occurrence/Uses |
|---|---|---|
Hydrochloric acid | HCl | Stomach acid, metal cleaning, food preparation |
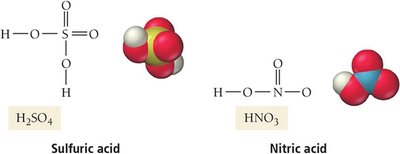
Sulfuric acid | H2SO4 | Fertilizer, batteries, dye production |
Nitric acid | HNO3 | Fertilizer, explosives, dyes |
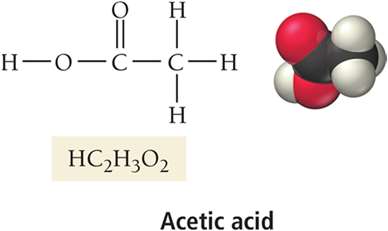
Acetic acid | HC2H3O2 | Vinegar, food preservation |
Citric acid | H3C6H5O7 | Citrus fruits, pH adjustment in foods |
Phosphoric acid | H3PO4 | Fertilizer, beverages, buffering |

Structures of Acids
Binary acids: H attached to a nonmetal (e.g., HCl, HF)
Oxyacids: H attached to an oxygen atom (e.g., H2SO4, HNO3)
Carboxylic acids: Contain the COOH group (e.g., acetic acid, citric acid)
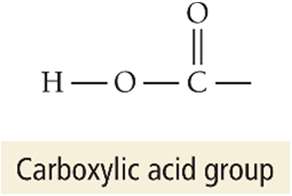
Properties and Examples of Bases
General Properties of Bases
Bitter taste (e.g., alkaloids in plants)
Slippery feel (e.g., soap)
Turn red litmus paper blue
Ability to neutralize acids

Common Bases and Their Uses
Name | Formula | Occurrence/Uses |
|---|---|---|
Sodium hydroxide | NaOH | Soap, plastic, petroleum processing |
Potassium hydroxide | KOH | Soap, batteries, cotton processing |
Sodium bicarbonate | NaHCO3 | Baking soda, antacid |
Ammonia | NH3 | Detergent, fertilizer, cleaning agent |
Definitions of Acids and Bases
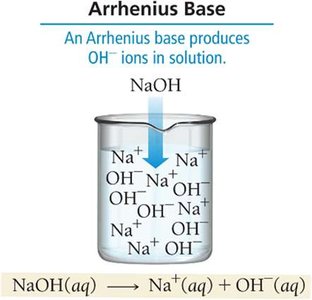
Arrhenius Definition
Acid: Produces H+ ions in aqueous solution
Base: Produces OH− ions in aqueous solution

Hydronium Ion Formation
In water, H+ ions associate with H2O to form the hydronium ion (H3O+):
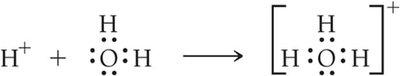
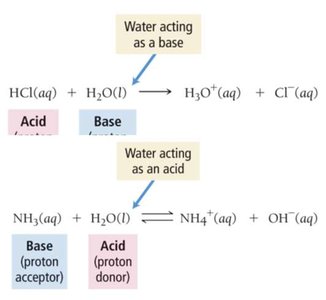
Brønsted–Lowry Definition
Acid: Proton (H+) donor
Base: Proton (H+) acceptor (must have a lone pair)
All Arrhenius acids/bases are also Brønsted–Lowry acids/bases, but the Brønsted–Lowry definition is broader and applies to more reactions.
Lewis Definition
Acid: Electron pair acceptor
Base: Electron pair donor
This definition is the most general and includes many reactions not covered by the other definitions.
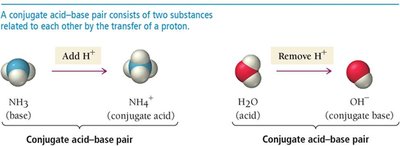
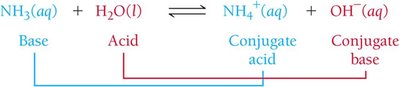
Acid–Base Reactions and Conjugate Pairs
Acid–Base Reactions
In an acid–base reaction, a proton is transferred from the acid to the base. The products are a conjugate base and a conjugate acid.
Conjugate acid: The species formed when a base gains a proton
Conjugate base: The species formed when an acid loses a proton
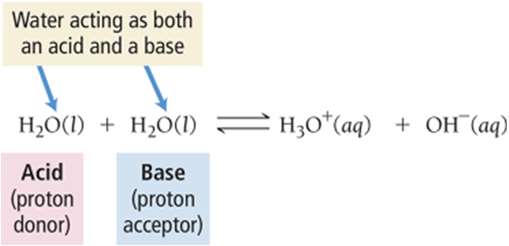
Amphoteric Substances
Amphoteric substances can act as either an acid or a base. Water is the most common example:
Strength of Acids and Bases
Strong vs. Weak Acids and Bases
Strong acids/bases: Completely ionize in water (strong electrolytes)
Weak acids/bases: Partially ionize in water (weak electrolytes)
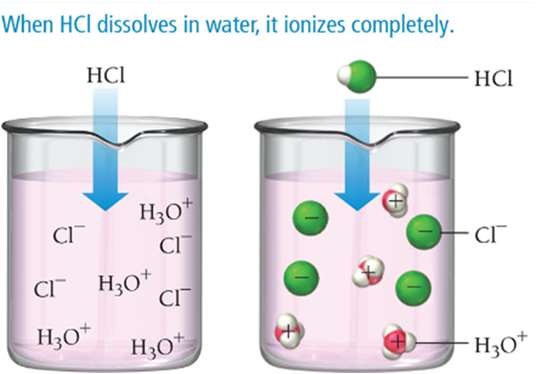
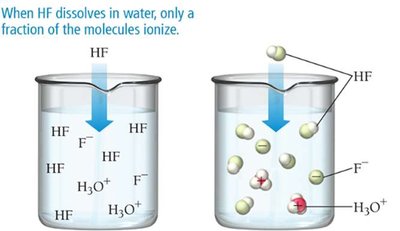
Examples of Strong and Weak Acids
Strong Acids | Weak Acids |
|---|---|
HCl, HNO3, H2SO4, HBr, HI, HClO4 | HF, H2CO3, H3PO4, CH3COOH |
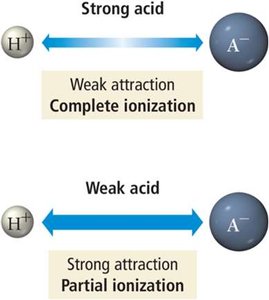
Acid Ionization Constant (Ka)
The strength of an acid is measured by its acid ionization constant, Ka:
Larger Ka = stronger acid; smaller Ka = weaker acid.
Autoionization of Water and the pH Scale
Autoionization of Water
Water can ionize to form hydronium and hydroxide ions:
The ion product constant for water is:
at 25°C
pH and pOH
pH = -\log[H_3O^+]
pOH = -\log[OH^-]
pH + pOH = 14.00 (at 25°C)
[H3O+] = 10^{-pH}
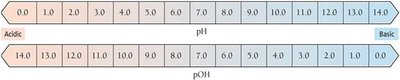
Interpreting pH Values
pH < 7: Acidic solution
pH = 7: Neutral solution
pH > 7: Basic solution
Each pH unit represents a tenfold change in [H3O+]
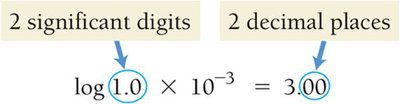
pKa and pKb
pKa = -\log Ka
pKb = -\log Kb
Stronger acids have smaller pKa values; stronger bases have smaller pKb values.
Sample Calculations
Given [H3O+] = 9.2 × 10−9 M, pH = −log(9.2 × 10−9) = 8.04
Given pH = 8.37, [H3O+] = 10−8.37 = 4.3 × 10−9 M
Summary Table: Acid–Base Concepts
Concept | Definition | Key Equation |
|---|---|---|
Arrhenius Acid | Produces H+ in water | HCl(aq) → H+(aq) + Cl−(aq) |
Arrhenius Base | Produces OH− in water | NaOH(aq) → Na+(aq) + OH−(aq) |
Brønsted–Lowry Acid | Proton donor | HA + H2O → A− + H3O+ |
Brønsted–Lowry Base | Proton acceptor | B + H2O → HB+ + OH− |
Lewis Acid | Electron pair acceptor | — |
Lewis Base | Electron pair donor | — |
