 Back
BackAcids, Bases, and Acid-Base Equilibria: Study Guide
Study Guide - Smart Notes

Acids and Bases: Definitions and Properties
Arrhenius and Brønsted-Lowry Definitions
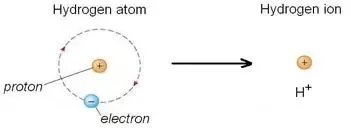
Acids and bases are fundamental chemical species with distinct behaviors in aqueous solutions. The Arrhenius definition states that acids produce H+ ions (or H3O+) in water, while bases produce OH- ions. The Brønsted-Lowry definition expands this concept: acids are proton (H+) donors, and bases are proton acceptors.
Arrhenius Acid: Increases [H+] in water.
Arrhenius Base: Increases [OH-] in water.
Brønsted-Lowry Acid: Donates a proton (H+).
Brønsted-Lowry Base: Accepts a proton (H+).
Example: NH3 + H2O ⇄ NH4+ + OH-
Acid and Base Properties
Acids: Sour taste, pH < 7, turn blue litmus paper red, react with carbonates.
Bases: Bitter taste, pH > 7, turn red litmus paper blue, react with wool.
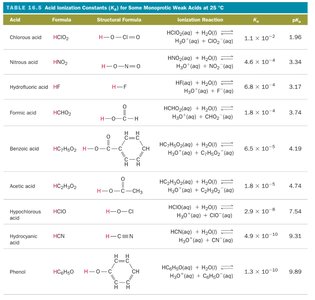
Strong and Weak Acids and Bases
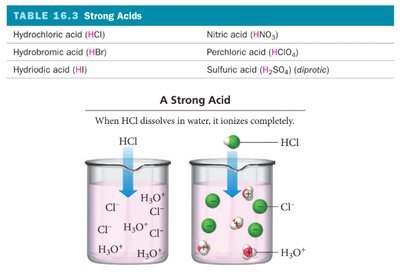
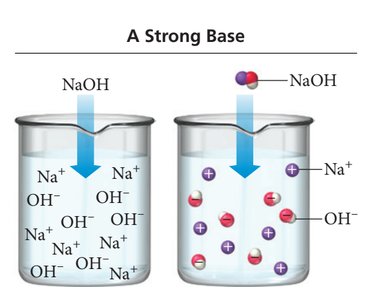
Strong Acids and Bases
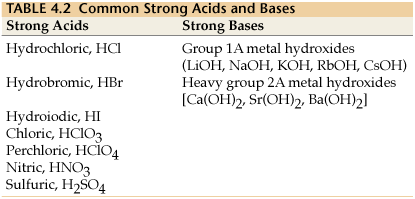
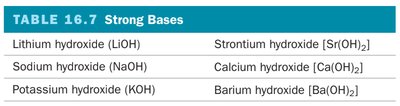
Strong acids and bases dissociate completely in water, resulting in high concentrations of ions. Common strong acids and bases are listed below:
Strong Acids | Strong Bases |
|---|---|
Hydrochloric acid (HCl) | Group 1A metal hydroxides (LiOH, NaOH, KOH, RbOH, CsOH) |
Hydrobromic acid (HBr) | Heavy group 2A metal hydroxides (Ca(OH)2, Sr(OH)2, Ba(OH)2) |
Hydroiodic acid (HI) | |
Chloric acid (HClO3) | |
Perchloric acid (HClO4) | |
Nitric acid (HNO3) | |
Sulfuric acid (H2SO4) |
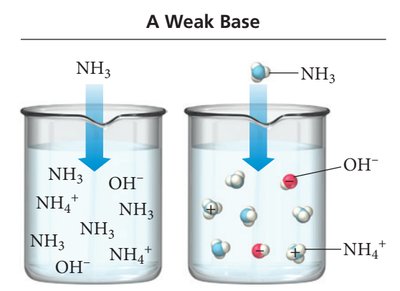
Weak Acids and Bases
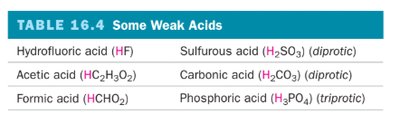
Weak acids and bases only partially dissociate in water, establishing an equilibrium between the undissociated and dissociated forms. Examples include acetic acid (HC2H3O2), formic acid (HCHO2), and ammonia (NH3).
Weak Acids |
|---|
Hydrofluoric acid (HF) |
Acetic acid (HC2H3O2) |
Formic acid (HCHO2) |
Sulfurous acid (H2SO3) |
Carbonic acid (H2CO3) |
Phosphoric acid (H3PO4) |
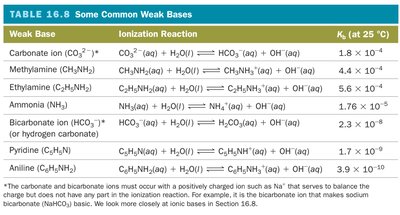
Weak Bases |
|---|
Ammonia (NH3) |
Methylamine (CH3NH2) |
Ethylamine (C2H5NH2) |
Pyridine (C5H5N) |
Aniline (C6H5NH2) |
Acid and Base Strength: Molecular and Particle Diagrams
Complete vs. Partial Ionization
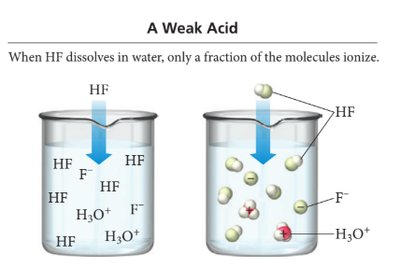
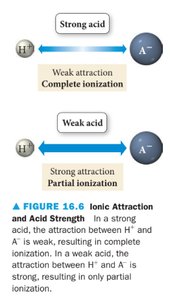
The strength of an acid or base depends on its degree of ionization. Strong acids and bases ionize completely, while weak acids and bases only partially ionize. The attraction between H+ and A- determines the extent of ionization.
Strong acid: Weak attraction between H+ and A- → complete ionization.
Weak acid: Strong attraction between H+ and A- → partial ionization.
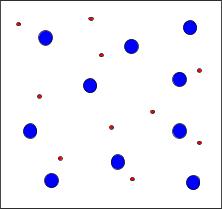
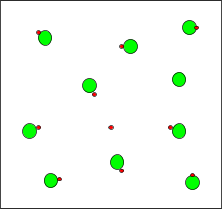
Particle Diagrams
Particle diagrams visually represent the relative amounts of undissociated molecules and ions in solution. For strong acids, most particles are ions; for weak acids, many remain as molecules.
Acid and Base Equilibria: Ka and Kb
Acid Ionization Constant (Ka)
The acid ionization constant, Ka, quantifies the strength of a weak acid. It is defined by the equilibrium:
HA(aq) + H2O(l) ⇄ H3O+(aq) + A-(aq)
Base Ionization Constant (Kb)
The base ionization constant, Kb, quantifies the strength of a weak base. It is defined by the equilibrium:
B(aq) + H2O(l) ⇄ BH+(aq) + OH-(aq)
Molecular Structure and Acid Strength
Binary Acids vs. Oxyacids
Acid strength is influenced by molecular structure. Binary acids (HX) depend on bond strength and ionic radius; oxyacids (H-O-X) depend on the number of oxygen atoms and electronegativity.
Binary acids: Larger ionic radius → weaker H-X bond → stronger acid.
Oxyacids: More oxygen atoms → greater electron withdrawal → stronger acid.
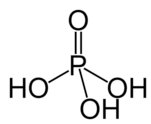
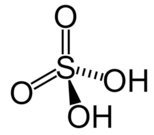
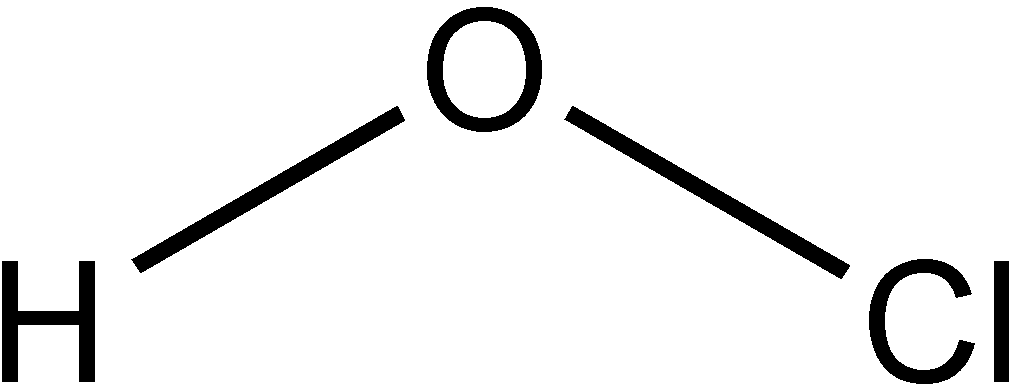
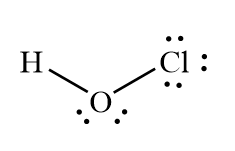
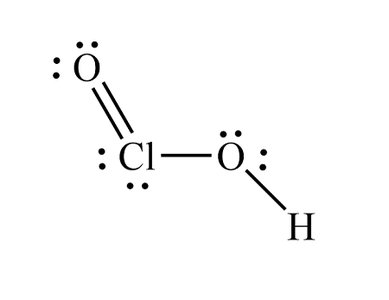
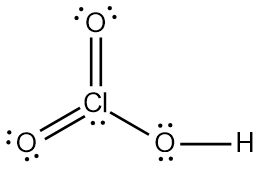
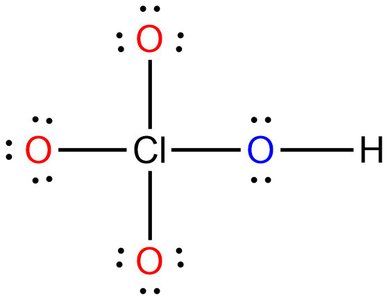
Trends in Acid Strength
Electronegativity and Acid Strength
For oxyacids, the more electronegative the central atom, the stronger the acid. For example, HOCl > HOBr > HOI in acid strength.
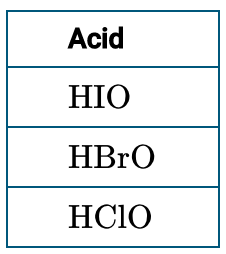
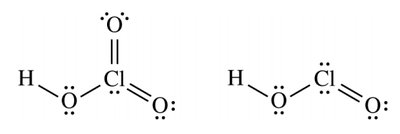
Number of Oxygens and Acid Strength
For acids like HOCl, HClO2, HClO3, and HClO4, acid strength increases with the number of oxygen atoms.
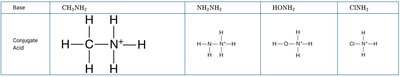
Conjugate Acids and Bases
Conjugate Pairs
Every acid-base reaction produces a conjugate acid and a conjugate base. The conjugate base is what remains after an acid donates a proton; the conjugate acid is what forms when a base accepts a proton.
Example: HCl + NH3 ⇄ Cl- + NH4+
Cl- is the conjugate base of HCl; NH4+ is the conjugate acid of NH3.
Acid-Base Equilibrium and pH
Autoionization of Water and pH Scale
Water can act as both an acid and a base (amphiprotic), and undergoes autoionization:
H2O(l) ⇄ H+(aq) + OH-(aq)
at 25°C
pH is defined as:
pOH is defined as:
Relationship:
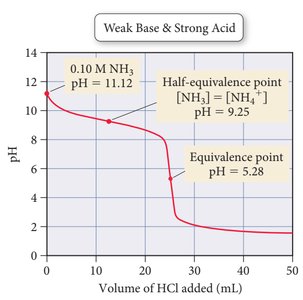
Acid and Base Titrations
Titration Principles
Titration is a laboratory technique used to determine the concentration of an acid or base by reacting it with a solution of known concentration. The equivalence point is reached when the moles of acid equal the moles of base.
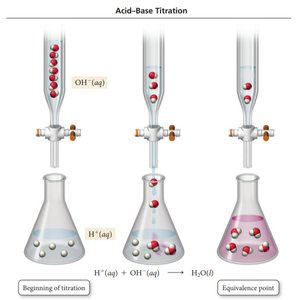
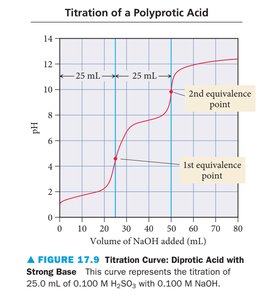
Titration Curves
Titration curves plot pH versus volume of titrant added. The shape of the curve depends on the strength of the acid and base involved.
Strong acid + strong base: Equivalence point at pH 7.
Weak acid + strong base: Equivalence point above pH 7.
Weak base + strong acid: Equivalence point below pH 7.
Polyprotic acid: Multiple equivalence points.
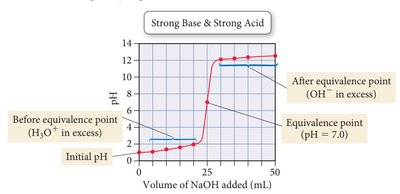
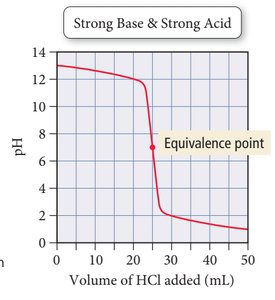
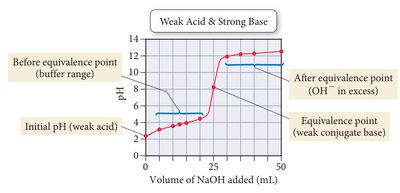


Indicators
Indicators are dyes that change color at specific pH ranges, used to signal the endpoint of a titration.
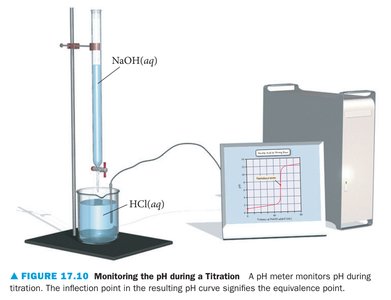
Buffer Solutions
Buffer Composition and Function
A buffer is a solution that resists changes in pH upon addition of small amounts of acid or base. Buffers contain significant amounts of a weak acid and its conjugate base, or a weak base and its conjugate acid.
Example: Acetic acid (HC2H3O2) and sodium acetate (NaC2H3O2).
Henderson-Hasselbalch Equation
The pH of a buffer can be calculated using the Henderson-Hasselbalch equation:
Buffer Capacity
Buffer capacity is the amount of acid or base a buffer can neutralize before the pH changes significantly. It increases with higher concentrations and more equal ratios of acid and base components.
Summary Table: Acid and Base Strength
Acid Strength | Base Strength |
|---|---|
Strong: HCl, H2SO4, HNO3, H3O+ | Strong: OH-, O2-, S2- |
Weak: HF, H2CO3, HCN, NH4+ | Weak: NH3, HCO3-, C2H3O2- |
Negligible: HS-, OH- | Negligible: HS-, OH- |
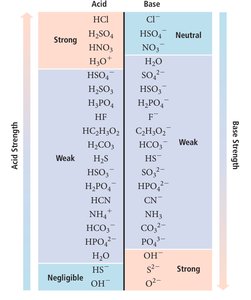
Key Equations and Relationships
at 25°C
Additional info:
Acid and base strength is determined by molecular structure, electronegativity, and the number of oxygen atoms in oxyacids.
Conjugate acid-base pairs are central to understanding equilibrium and buffer systems.
Titration curves and indicators are essential for laboratory determination of unknown concentrations.
