 Back
BackAcids, Bases, and Aqueous Equilibria: Study Notes for General Chemistry
Study Guide - Smart Notes

Acids and Bases: Fundamental Concepts
Introduction to Acids and Bases
Acids and bases are two fundamental classes of compounds in chemistry, characterized by their distinct properties and behaviors in aqueous solutions. Understanding their definitions, properties, and reactions is essential for mastering general chemistry.
Acids are substances that can donate protons (H+) or produce hydronium ions (H3O+) in water.
Bases are substances that can accept protons or produce hydroxide ions (OH-) in water.
The pH scale is used to quantify the acidity or basicity of a solution, ranging from 0 (most acidic) to 14 (most basic), with 7 being neutral.
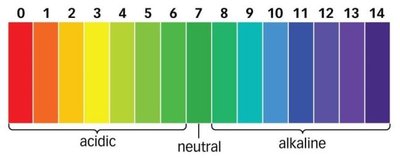
Properties and Examples of Acids and Bases
Acids typically have a sour taste, can dissolve metals, turn blue litmus paper red, and neutralize bases.
Bases usually have a bitter taste, slippery feel, turn red litmus paper blue, and neutralize acids.
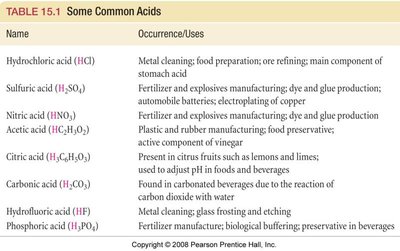
Common examples and their uses are summarized below:
Name | Occurrence/Uses |
|---|---|
Hydrochloric acid (HCl) | Metal cleaning; food preparation; ore refining; main component of stomach acid |
Sulfuric acid (H2SO4) | Fertilizer and explosives manufacturing; dye and glue production; automobile batteries; electroplating of copper |
Nitric acid (HNO3) | Fertilizer and explosives manufacturing; dye and glue production |
Acetic acid (HC2H3O2) | Plastic and rubber manufacturing; food preservative; active component of vinegar |
Citric acid (H3C6H5O7) | Present in citrus fruits such as lemons and limes |
Carbonic acid (H2CO3) | Found in carbonated beverages due to the reaction of carbon dioxide with water |
Hydrofluoric acid (HF) | Metal cleaning; glass frosting and etching |
Phosphoric acid (H3PO4) | Fertilizer manufacture; biological buffering; preservative in beverages |
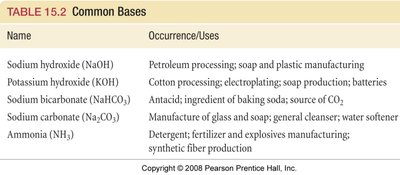
Name | Occurrence/Uses |
|---|---|
Sodium hydroxide (NaOH) | Petroleum processing; soap and plastic manufacturing |
Potassium hydroxide (KOH) | Cotton processing; electroplating; soap production; batteries |
Sodium bicarbonate (NaHCO3) | Antacid; ingredient of baking soda; source of CO2 |
Sodium carbonate (Na2CO3) | Manufacture of glass and soap; general cleanser; water softener |
Ammonia (NH3) | Detergent; fertilizer and explosives manufacturing; synthetic fiber production |
pH, pOH, and Calculations
The pH and pOH Scales
The pH and pOH scales are logarithmic measures of the concentrations of hydronium and hydroxide ions, respectively. They are related by the ionization constant of water, Kw.
pH is defined as
pOH is defined as
The relationship between pH and pOH at 25°C is
The ion product of water: at 25°C
![Diagram showing the relationships between pH, pOH, [H+], and [OH-]](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/a0d79ae3_image_4.png)
Logarithmic Calculations and Significant Figures
When taking the logarithm of a number with N significant figures, the result should have N decimal places.
When raising 10 to a power with N decimal places, the result should have N significant figures.
Practice Problems: Calculating pH and pOH
Practice problems often test your understanding of the relationships and calculations involving pH and pOH.


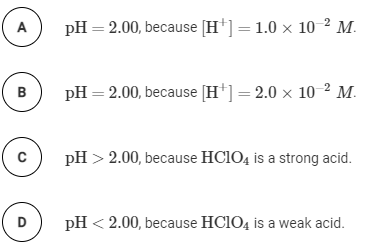
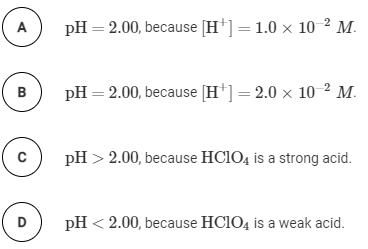
Comparing Acid Strengths
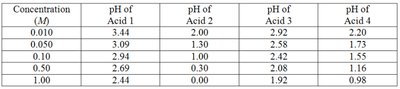
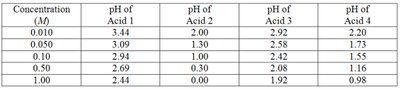
The strength of an acid is determined by its degree of ionization in water. Strong acids completely ionize, while weak acids only partially ionize. The table below shows experimental pH values for different acids at various concentrations, which can be used to identify strong and weak acids.
Concentration (M) | pH of Acid 1 | pH of Acid 2 | pH of Acid 3 | pH of Acid 4 |
|---|---|---|---|---|
0.010 | 3.44 | 2.00 | 2.92 | 2.20 |
0.050 | 3.09 | 1.30 | 2.58 | 1.73 |
0.10 | 2.94 | 1.00 | 2.42 | 1.55 |
0.50 | 2.69 | 0.30 | 2.08 | 1.16 |
1.00 | 2.44 | 0.00 | 1.92 | 0.98 |
Acid and Base Strength: Ka and pKa
Acid Dissociation Constant (Ka) and pKa
The strength of a weak acid is quantified by its acid dissociation constant, Ka, and its logarithmic counterpart, pKa:
The smaller the Ka, the weaker the acid; the smaller the pKa, the stronger the acid.

Acid-Base Equilibria and Buffer Solutions
Buffer Solutions and Their Properties
A buffer is a solution that resists changes in pH upon the addition of small amounts of acid or base. Buffers are typically made from a weak acid and its conjugate base, or a weak base and its conjugate acid.
Buffers work by neutralizing added H+ or OH- ions.
The Henderson-Hasselbalch equation is used to estimate the pH of a buffer:
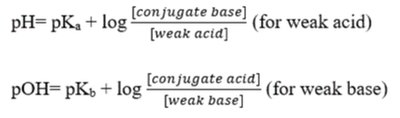
Acid-Base Titrations
Titration is a laboratory technique used to determine the concentration of an unknown acid or base by reacting it with a solution of known concentration (the titrant). The point at which the reaction is complete is called the equivalence point.
Monoprotic acids donate one proton per molecule; their titration curves have one equivalence point.
Polyprotic acids donate more than one proton; their titration curves have multiple equivalence points.
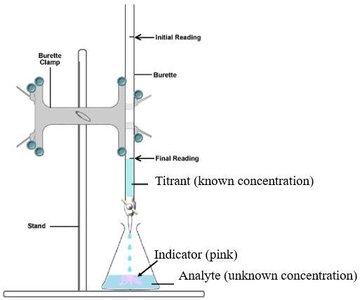
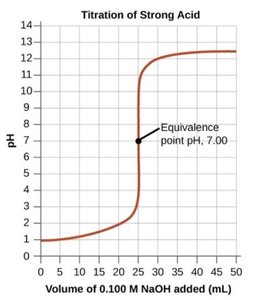
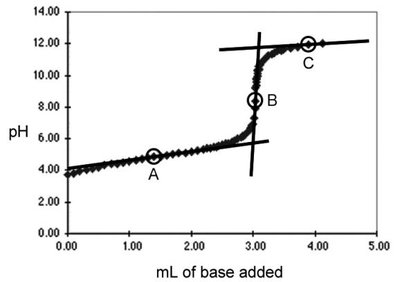
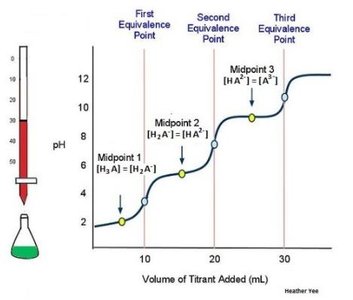
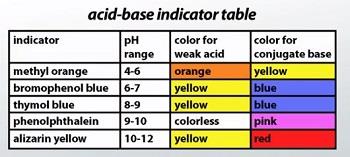
Acid-Base Indicators
Indicators and Their pH Ranges
Indicators are substances that change color depending on the pH of the solution. They are used to determine the endpoint of titrations. Each indicator has a specific pH range over which it changes color.
Indicator | pH Range | Color for Weak Acid | Color for Conjugate Base |
|---|---|---|---|
Methyl orange | 4-6 | orange | yellow |
Bromophenol blue | 6-7 | yellow | blue |
Thymol blue | 8-9 | yellow | blue |
Phenolphthalein | 9-10 | colorless | pink |
Alizarin yellow | 10-12 | yellow | red |
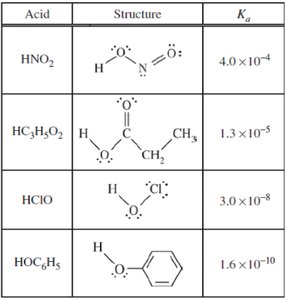
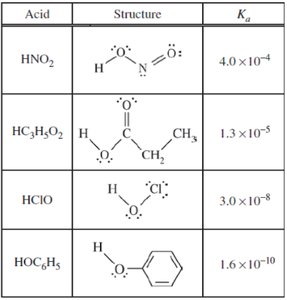
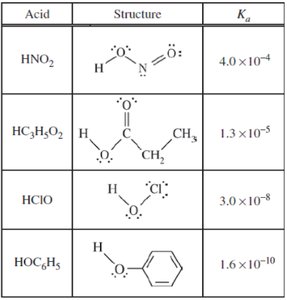
Summary Table: Acid Dissociation Constants
The table below summarizes the structures and Ka values for several weak acids, which are important for buffer calculations and understanding acid strength.
Acid | Structure | Ka |
|---|---|---|
HNO2 | Structure shown | 4.0 × 10-4 |
HC3H5O2 | Structure shown | 1.3 × 10-5 |
HClO | Structure shown | 3.0 × 10-8 |
HOC6H5 | Structure shown | 1.6 × 10-10 |
Additional info: These notes cover the essential concepts of acids, bases, pH, pOH, buffer solutions, titrations, and indicators, as well as the mathematical relationships and laboratory applications relevant to a general chemistry course.
