 Back
BackAcids, Bases, and Buffers: General Chemistry Study Notes
Study Guide - Smart Notes

Acids and Bases
Introduction to Acids and Bases
Acids and bases are fundamental chemical species that play a crucial role in many chemical reactions and biological processes. Acids are typically characterized by their sour taste, while bases are known for their bitter or salty taste.
Acids: Substances that produce H3O+ (hydronium ion) in aqueous solution.
Bases: Substances that produce OH- (hydroxide ion) in aqueous solution.



Arrhenius Definition
The Arrhenius definition is one of the earliest and simplest ways to classify acids and bases.
Acid: Produces H+ (or H3O+) ions in water.
Base: Produces OH- ions in water.
Brønsted-Lowry Definition
The Brønsted-Lowry definition expands the concept of acids and bases beyond aqueous solutions.
Acid: Proton (H+) donor.
Base: Proton (H+) acceptor.
Conjugate acid-base pair: When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid.
Examples of Acid-Base Reactions
CH3COOH(aq) + H2O(l) → CH3COO-(aq) + H3O+(aq)
NH3(aq) + H2O(l) → NH4+(aq) + OH-(aq)
Strong vs. Weak Acids and Bases
Strong acids/bases: Completely ionize in aqueous solution.
Weak acids/bases: Partially ionize in aqueous solution.
Electrolytes
Electrolytes are substances that conduct electricity when dissolved in water.
Strong electrolytes: Completely dissociate into ions (e.g., NaCl).
Weak electrolytes: Partially dissociate (e.g., CH3COOH).
Nonelectrolytes: Do not dissociate (e.g., distilled water).
Acid and Base Strength
A strong acid contains a weak conjugate base.
A strong base contains a weak conjugate acid.
Types of Acids
Monoprotic acids: Donate one proton (e.g., HCl).
Diprotic acids: Donate two protons (e.g., H2SO4).
Triprotic acids: Donate three protons (e.g., H3PO4).
Amphiprotic: Can act as either an acid or a base (e.g., H2O).
Oxyacids and Organic Acids
Oxyacids: Acidic hydrogen is attached to an oxygen atom (e.g., H2SO4, H3PO4).
Organic acids: Contain a carboxyl group (-COOH), usually weak (e.g., CH3COOH).
Naming Acids
Binary acids: Hydro + base name of nonmetal + -ic acid (e.g., Hydrochloric acid).
Ternary acids: Based on polyatomic ions: -ate ion → -ic acid, -ite ion → -ous acid (e.g., Nitric acid from nitrate, Nitrous acid from nitrite).
Acid Ionization and pH
Acid Ionization Constant (Ka)
The acid ionization constant, Ka, measures the strength of an acid in solution.
For strong acids, Ka is not used because they ionize completely.
pKa is related to Ka:
Stronger acids have higher Ka and lower pKa.
Ionization of Water and pH Scale
Water can ionize to form hydronium and hydroxide ions.
at 25°C
Neutral solution:
Acidic solution:
Basic solution:
pH and pOH Calculations

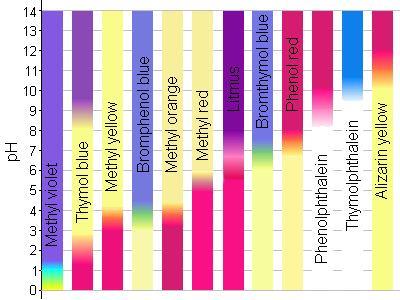

pH Indicators in Nature
Some plants and flowers change color depending on the pH of the soil.
Bigleaf hydrangea flowers are pink in acidic soil and blue in basic (alkaline) soil.


Acid-Base Reactions and Neutralization
Neutralization Reactions
Neutralization occurs when an acid reacts with a base to produce a salt and water.
General equation: Acid + Base → Salt + Water
Example:
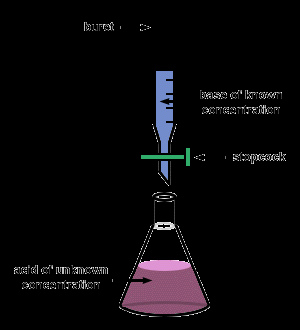
Titration
Titration is a laboratory technique used to determine the concentration of an unknown acid or base by reacting it with a solution of known concentration.
Equivalence point: The point at which equal amounts of acid and base have reacted (pH = 7 for strong acid-strong base titrations).
Calculation: (for monoprotic acids and bases)
Buffers
Buffer Solutions
Buffers are solutions that resist changes in pH when small amounts of acid or base are added.
Composed of a weak acid and its conjugate base, or a weak base and its conjugate acid.
Example: Acetic acid (CH3COOH) and sodium acetate (CH3COO-Na+).




Buffer Composition and Types
Carbonate buffer: H2CO3 / HCO3-
Phosphate buffer: H2PO4- / HPO42-
Protein buffer: Proteins can act as buffers in biological systems.
Buffer Action
Buffers maintain pH by neutralizing added acids or bases.
When acid is added:
When base is added:


Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation relates the pH of a buffer solution to the concentration of the acid and its conjugate base.
Used to calculate the pH of buffer solutions.
Summary Table: Acid, Base, and Buffer Properties
Type | Definition | Example |
|---|---|---|
Acid | Donates H+ | HCl, CH3COOH |
Base | Accepts H+ | NaOH, NH3 |
Buffer | Resists pH change | CH3COOH/CH3COO-, H2CO3/HCO3- |
Additional info:
Blood is a natural buffer, maintaining pH between 7.35 and 7.45.
pH indicators are used in laboratory and nature to visually determine pH.
