 Back
BackAcids, Bases, and Coordination Chemistry: General Chemistry II Study Guide
Study Guide - Smart Notes

Acid-Base Theories and Concepts
Arrhenius and Brønsted-Lowry Theories
The Arrhenius and Brønsted-Lowry theories provide foundational definitions for acids and bases. The Arrhenius theory states that acids are substances that produce H+ ions in aqueous solution, while bases produce OH- ions. The Brønsted-Lowry theory expands this definition: acids are proton (H+) donors, and bases are proton acceptors.
Arrhenius Acid: Produces H+ in water.
Arrhenius Base: Produces OH- in water.
Brønsted-Lowry Acid: Donates a proton (H+).
Brønsted-Lowry Base: Accepts a proton (H+).
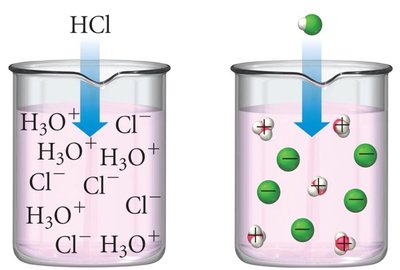
Example: HCl(aq) + H2O(l) → Cl-(aq) + H3O+(aq)

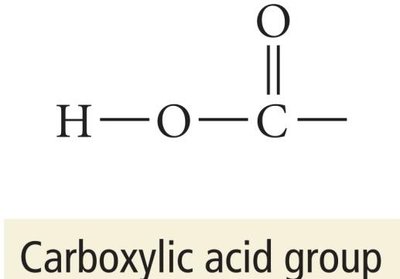
Types of Acids
Acids are classified based on their structure and the number of ionizable hydrogens:
Binary acids: Composed of hydrogen and one other element (e.g., HCl).
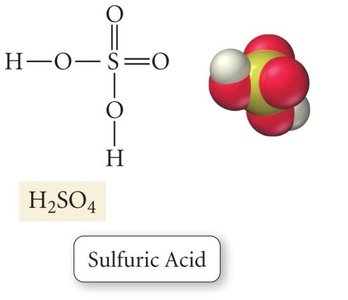
Ternary/oxoacids: Contain hydrogen, oxygen, and another element (e.g., H2SO4).
Polyprotic acids: Have more than one ionizable hydrogen (e.g., H2SO4, H3PO4).
Carboxylic acids: Contain the carboxyl group (-COOH).
Acid and Base Strength
Strong vs. Weak Acids and Bases
The strength of an acid or base depends on its degree of ionization in water. Strong acids and bases ionize completely, while weak acids and bases only partially ionize.
Strong acids: 100% ionized in water; strong electrolytes.
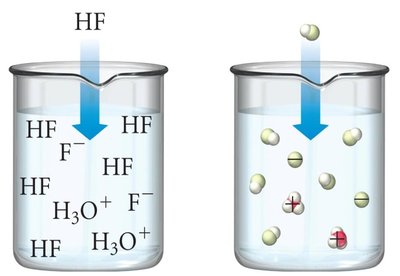
Weak acids: Less than 1% ionized; weak electrolytes.
Strong bases: Group I and II hydroxides (except Be and Mg); strong electrolytes.
Weak bases: Only a small fraction accept protons; weak electrolytes.
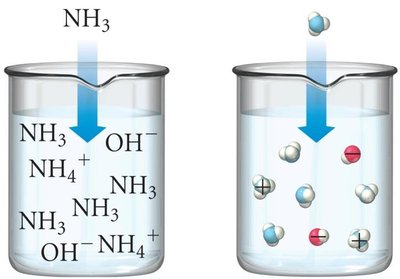
Conjugate Acid-Base Pairs
Identifying Conjugate Pairs
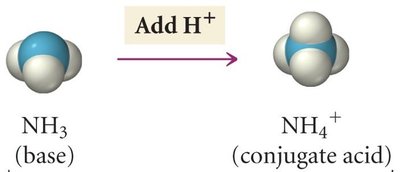
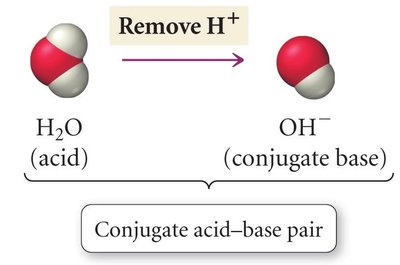
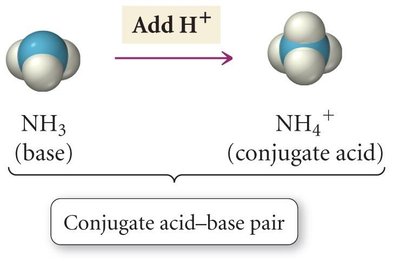
Conjugate acid-base pairs differ by one proton. When an acid donates a proton, it forms its conjugate base; when a base accepts a proton, it forms its conjugate acid.
Acid/Conjugate Base Pair: H2O and OH-
Base/Conjugate Acid Pair: NH3 and NH4+
Example: NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq)
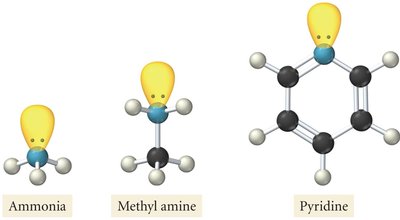
Structure and Basicity of Amines
Lewis Structures and Basicity
Amines are derivatives of ammonia (NH3) where one or more hydrogen atoms are replaced by hydrocarbon groups. The lone pair of electrons on the nitrogen atom is responsible for the basicity of amines, allowing them to accept protons.
Primary amine: One hydrogen replaced.
Secondary amine: Two hydrogens replaced.
Tertiary amine: All three hydrogens replaced.
Example: Methylamine (CH3NH2), Pyridine (C5H5N)
Autoionization of Water and pH Calculations
Autoionization and Ion Product (Kw)
Water undergoes autoionization, producing equal concentrations of H3O+ and OH- ions. The ion product of water (Kw) is a constant at 25°C.
Autoionization equation:
Ion product:
Neutral water:
pH and pOH Calculations
The pH of a solution is calculated using the concentration of hydronium ions. pOH is similarly calculated for hydroxide ions. The sum of pH and pOH is always 14 at 25°C.
pH formula:
pOH formula:
Relationship:
Hydronium ion concentration:
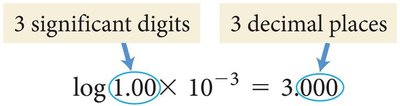
Equilibrium Calculations for Weak Acids and Bases
Acid Dissociation Constant (Ka) and Base Dissociation Constant (Kb)
Weak acids and bases do not fully dissociate in water. Their equilibrium concentrations are calculated using the acid dissociation constant (Ka) or base dissociation constant (Kb).
Acid dissociation:
Equilibrium expression:
Base dissociation:
Equilibrium expression:
R.I.C.E. Charts
R.I.C.E. charts (Reaction, Initial, Change, Equilibrium) are used to organize and solve equilibrium problems for weak acids and bases.
Step 1: Write the balanced equation.
Step 2: Fill in initial concentrations.
Step 3: Define the change (x).
Step 4: Write equilibrium concentrations.
Step 5: Substitute into equilibrium expression and solve for x.
Acidic, Basic, and Neutral Salt Solutions
Hydrolysis of Salts
Salts can produce acidic, basic, or neutral solutions depending on the nature of their ions. Hydrolysis reactions occur when ions react with water to produce H3O+ or OH-.
Basic salts: Contain conjugate bases of weak acids.
Acidic salts: Contain conjugate acids of weak bases.
Neutral salts: Contain ions that do not react with water.
Example: NH4Cl produces an acidic solution; NaF produces a basic solution.
Polyprotic Acids and Acid Strength
Polyprotic Acids
Polyprotic acids have more than one ionizable hydrogen and ionize in steps, each with its own Ka value. The first ionization is usually the strongest.
Monoprotic: One ionizable H (e.g., HCl).
Diprotic: Two ionizable H (e.g., H2SO4).
Triprotic: Three ionizable H (e.g., H3PO4).
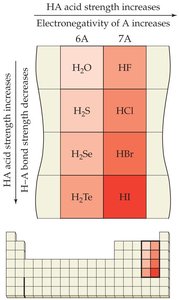
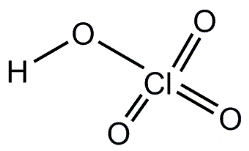
Factors Affecting Acid Strength
The strength of binary and oxoacids depends on bond polarity, bond strength, electronegativity, and the number of oxygen atoms attached to the central atom.
Binary acids: Strength increases with electronegativity and decreases with bond strength.
Oxoacids: Strength increases with the number of oxygen atoms and the electronegativity of the central atom.
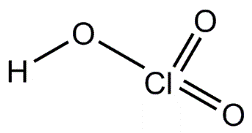
Lewis Acid-Base Theory and Coordination Chemistry
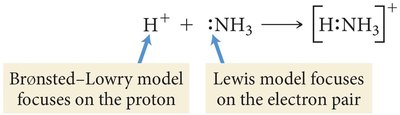
Lewis Acids and Bases
The Lewis theory defines acids as electron pair acceptors and bases as electron pair donors. This theory is especially important in coordination chemistry.
Lewis acid: Electron pair acceptor (e.g., metal cations).
Lewis base: Electron pair donor (e.g., ligands with lone pairs).
Adduct: Product formed when a Lewis base donates electrons to a Lewis acid.
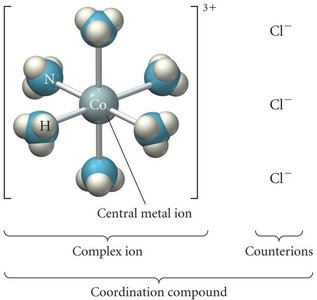
Coordination Compounds
Coordination compounds consist of a central metal ion surrounded by ligands. The coordination number is the number of ligand attachments to the metal center.
Complex ion: Metal ion bonded to ligands.
Coordination compound: Complex ion plus counterions.
Common coordination numbers: 4 (tetrahedral), 6 (octahedral).
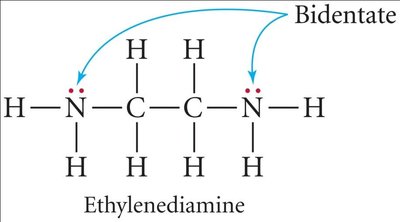
Ligands and Chelation
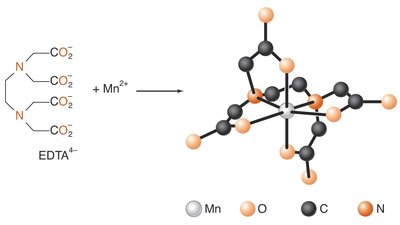
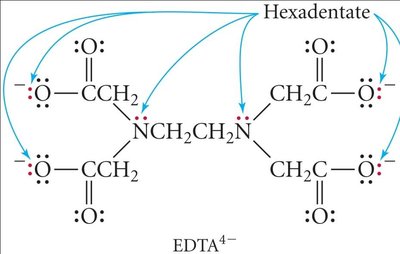
Ligands can be monodentate (one binding site), bidentate (two binding sites), or polydentate (multiple binding sites). Chelation occurs when a polydentate ligand forms a ring structure with the metal ion.
Monodentate: One donor atom (e.g., NH3).
Bidentate: Two donor atoms (e.g., ethylenediamine).
Polydentate: Multiple donor atoms (e.g., EDTA).
Naming Coordination Compounds
Coordination compounds are named by listing ligands alphabetically, followed by the metal and its oxidation state. Anionic complex ions use the Latin root and end in "-ate".
Ligand names: Anionic ligands end in "o"; neutral ligands have specific names (e.g., aqua, ammine).
Prefixes: di-, tri-, tetra- for number of ligands; bis-, tris-, tetrakis- for polydentate ligands.
Example: Potassium amminepentachloridoplatinate(IV) for K[Pt(NH3)Cl5]
![[Pt(NH3)Cl5]- structure](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/062b713b_image_36.png)
Coordination Geometry
The geometry of coordination compounds depends on the metal ion and the number of ligands. Common geometries include octahedral, tetrahedral, square planar, and linear.
Octahedral: 6 ligands (e.g., [Co(NH3)6]3+)
Tetrahedral: 4 ligands (e.g., Al3+, Zn2+)
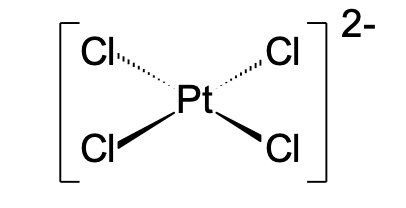
Square planar: d8 ions (e.g., Pt2+, Cu2+)
Linear: Ag+ (rare)
Summary Table: Common Weak Bases
The following table summarizes some common weak bases, their ionization reactions, and Kb values at 25°C.
Weak Base | Ionization Reaction | Kb (at 25°C) |
|---|---|---|
Carbonate Ion (CO32-) | CO32-(aq) + H2O(l) ⇌ HCO3-(aq) + OH-(aq) | 1.8 × 10-4 |
Methylamine (CH3NH2) | CH3NH2(aq) + H2O(l) ⇌ CH3NH3+(aq) + OH-(aq) | 4.4 × 10-4 |
Ethylamine (C2H5NH2) | C2H5NH2(aq) + H2O(l) ⇌ C2H5NH3+(aq) + OH-(aq) | 5.6 × 10-4 |
Ammonia (NH3) | NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH-(aq) | 1.76 × 10-5 |
Bicarbonate Ion (HCO3-) | HCO3-(aq) + H2O(l) ⇌ H2CO3(aq) + OH-(aq) | 1.7 × 10-10 |
Pyridine (C5H5N) | C5H5N(aq) + H2O(l) ⇌ C5H5NH+(aq) + OH-(aq) | 1.7 × 10-9 |
Aniline (C6H5NH2) | C6H5NH2(aq) + H2O(l) ⇌ C6H5NH3+(aq) + OH-(aq) | 3.9 × 10-10 |
Additional info:
Some explanations and examples were expanded for clarity and completeness, including the use of R.I.C.E. charts, naming conventions for coordination compounds, and the summary table of weak bases.
