 Back
BackAcids, Bases, and Equilibrium: Brønsted–Lowry Theory and Calculations
Study Guide - Smart Notes

Acids and Bases: Brønsted–Lowry Theory
Identifying Acids, Bases, and Their Conjugates
The Brønsted–Lowry theory defines acids as proton donors and bases as proton acceptors. In aqueous reactions, each acid or base forms a conjugate pair after transferring a proton.
Acid: Donates a proton (H+) to another species.
Base: Accepts a proton.
Conjugate Acid: The species formed when a base gains a proton.
Conjugate Base: The species formed when an acid loses a proton.
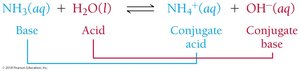
Example: In the reaction NH3(aq) + H2O(l) → NH4+(aq) + OH–(aq), NH3 is the base, H2O is the acid, NH4+ is the conjugate acid, and OH– is the conjugate base.
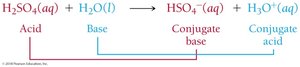
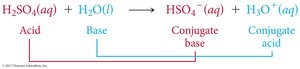
Example: In the reaction H2SO4(aq) + H2O(l) → HSO4–(aq) + H3O+(aq), H2SO4 is the acid, H2O is the base, HSO4– is the conjugate base, and H3O+ is the conjugate acid.
Calculating Ion Concentrations and Solution Classification
Using the Ion Product Constant (Kw)
The ion product constant for water, Kw, is used to relate the concentrations of hydronium ions ([H3O+]) and hydroxide ions ([OH–]) in aqueous solutions:
Formula:
Acidic Solution: [H3O+] > [OH–]
Basic Solution: [H3O+] < [OH–]
Neutral Solution: [H3O+] = [OH–]
![Calculation of [OH-] for acidic solution](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/564f73f1_image_6.png)
![Calculation of [OH-] for basic solution](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/564f73f1_image_7.png)
![Calculation of [OH-] for neutral solution](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/564f73f1_image_8.png)
Calculating pH and pOH
pH from Hydronium Ion Concentration
The pH of a solution is calculated using the concentration of hydronium ions:
Formula:
Acidic: pH < 7
Basic: pH > 7
Example: For [H3O+] = 1.8 × 10–4 M, pH = 3.74 (acidic).
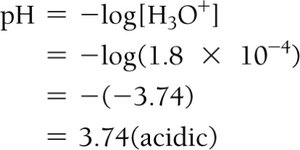
Example: For [OH–] = 1.3 × 10–2 M, first calculate [H3O+] using Kw, then pH = 12.11 (basic).
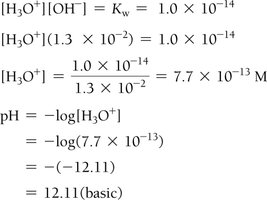
Equilibrium Calculations for Weak Acids and Bases
ICE Tables and Equilibrium Expressions
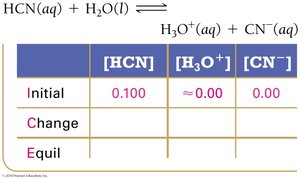
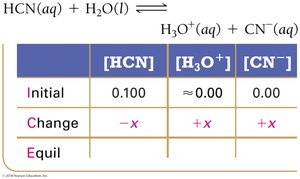
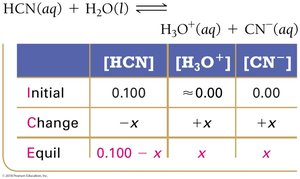
To solve equilibrium problems for weak acids and bases, use ICE tables (Initial, Change, Equilibrium) and equilibrium constant expressions.
Step 1: Write the balanced equation and set up the ICE table.
Step 2: Represent changes in concentration with a variable (usually x).
Step 3: Sum columns to find equilibrium concentrations.
Step 4: Substitute equilibrium concentrations into the Ka or Kb expression.
Step 5: Solve for x, check the validity of the 'x is small' approximation.
Step 6: Calculate pH or percent ionization as needed.
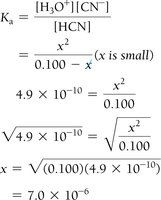
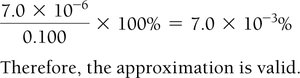
Acid Ionization Constants and Tables
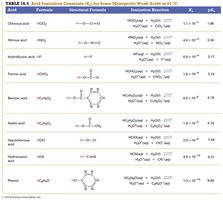
Acid ionization constants (Ka) quantify the strength of weak acids. Reference tables provide Ka and pKa values for common acids.
Ka:
pKa:
Example Table: See below for typical values.
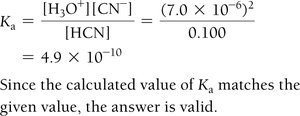
Summary Table: Strong Acids
Strong acids dissociate completely in water. Common strong acids include:
Acid | Formula |
|---|---|
Hydrochloric acid | HCl |
Hydrobromic acid | HBr |
Hydroiodic acid | HI |
Nitric acid | HNO3 |
Perchloric acid | HClO4 |
Sulfuric acid (diprotic) | H2SO4 |
Summary Table: Acid Ionization Constants for Weak Acids
This table lists Ka and pKa values for common weak acids at 25°C.
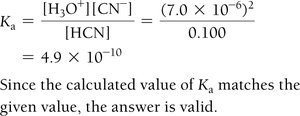
Example: ICE Table for Weak Base (NH3)
For the weak base NH3, the ICE table helps determine equilibrium concentrations and calculate Kb.
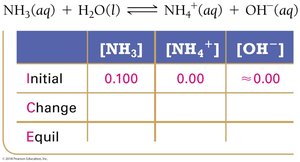
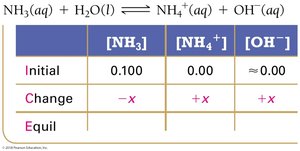
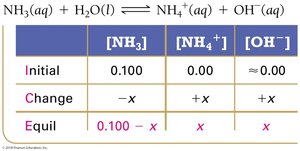
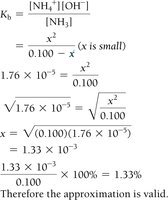
Key Equations
Ion Product Constant:
pH:
Ka:
Kb:
Percent Ionization:
Additional info: The notes above expand on the original content by providing definitions, formulas, and context for ICE tables, equilibrium calculations, and acid/base classification. Tables are recreated for clarity and completeness.
