 Back
BackAcids, Bases, and Salt Preparations: Study Notes for General Chemistry
Study Guide - Smart Notes

Solubility Rules
Solubility of Ionic Compounds
Ionic compounds are generally more soluble in water than covalent substances, but there are important exceptions. Understanding solubility rules is essential for predicting the outcomes of reactions and for preparing salts in the laboratory.
Sodium, potassium, and ammonium salts: All are soluble.
Nitrates: All are soluble.
Chlorides: Most are soluble, except silver and lead(II) chlorides.
Sulfates: Most are soluble, except barium, calcium, and lead(II) sulfates.
Carbonates: Only sodium, potassium, and ammonium carbonates are soluble; most others are insoluble.
Hydroxides: Only sodium, potassium, and ammonium hydroxides are soluble; calcium hydroxide is sparingly soluble, most others are insoluble.
Example: Calcium hydroxide solution (limewater) is used to test for carbon dioxide.
Acids, Bases & Protons
Proton Transfer Theory
The modern definitions of acids and bases are based on their behavior with protons (H+ ions).
Acids: Proton donors; they ionize in solution to produce H+ ions, making the solution acidic. Example:
Bases (Alkalis): Proton acceptors; they ionize in solution to produce OH- ions, which can accept protons and make the solution alkaline. Example:
Reactions of Acids
Acid Reactions with Metals
Only metals above hydrogen in the reactivity series react with dilute acids, forming a salt and hydrogen gas. Highly reactive metals (e.g., potassium, sodium) react explosively.
General equation:
Example:
Acid Reactions with Bases (Neutralization)
Acids react with bases (metal oxides or hydroxides) to produce a salt and water.
General equation:
Example:
Acid Reactions with Metal Carbonates
Acids react with metal carbonates to produce a salt, carbon dioxide, and water. Effervescence (bubbling) is observed due to CO2 gas.
General equation:
Example:
Bases
Definition and Properties
Bases neutralize acids, forming a salt and water. Not all bases are alkalis; only water-soluble bases are called alkalis. Alkalis have pH values above 7 and turn red litmus paper blue.
Common bases: Metal oxides, hydroxides, and carbonates.
Alkalis: Sodium hydroxide (NaOH), potassium hydroxide (KOH), aqueous ammonia (NH3 + H2O).
Example: Ammonia gas dissolves in water to form ammonium hydroxide.
Preparation of Salts
Preparation of Soluble Salts (Insoluble Base Method)
Soluble salts can be prepared by reacting an acid with an insoluble base (oxide, hydroxide, or carbonate). The base is added in excess to ensure all acid reacts, then filtered, and the solution is evaporated to crystallize the salt.
Example:
Result: Hydrated copper(II) sulfate crystals are bright blue and regularly shaped.

Preparation of Soluble Salts (Titration Method)
A titration can be used to prepare a dry salt from an acid and an alkali. The alkali is measured into a flask, indicator added, and acid is titrated in until neutralization. The same volumes are mixed without indicator, evaporated, and crystallized.
Key steps: Use pipette and burette for accurate measurement, add indicator, titrate, evaporate, and crystallize.
Preparation of Insoluble Salts (Precipitation Method)
Insoluble salts are prepared by mixing two soluble salt solutions, resulting in a precipitate. The precipitate is filtered, washed, and dried.
General equation:
Example:
Step-by-Step Process for Preparing an Insoluble Salt
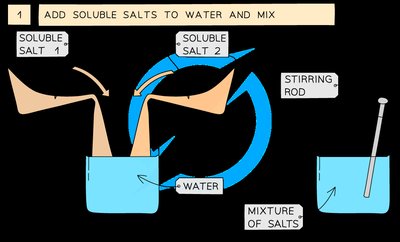
Add soluble salts to water and mix thoroughly with a stirring rod.
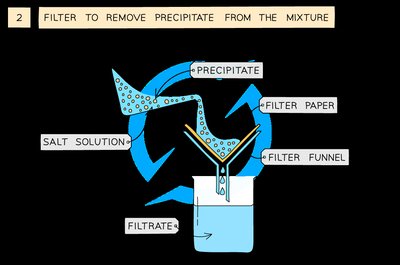
Filter the mixture to remove the precipitate using filter paper and funnel.
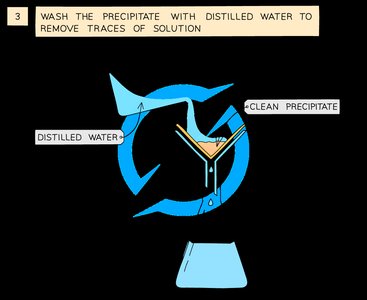
Wash the precipitate with distilled water to remove traces of solution.
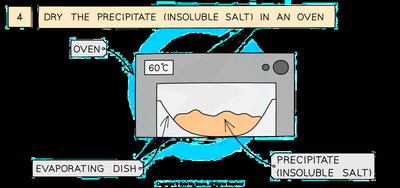
Dry the precipitate (insoluble salt) in an oven at 60°C using an evaporating dish.
Practical Examples
Preparation of Copper(II) Sulfate
React copper(II) oxide with dilute sulfuric acid, filter, evaporate, and crystallize to obtain hydrated copper(II) sulfate crystals.
Preparation of Lead(II) Sulfate
Mix lead(II) nitrate and potassium sulfate solutions, filter the precipitate, wash, and dry to obtain lead(II) sulfate.
Key Laboratory Apparatus
Beaker, spatula, glass rod, measuring cylinder, Bunsen burner, tripod, gauze, heatproof mat, filter funnel, filter paper, conical flask, evaporating basin, oven.
Examiner Tips
Always add the solid base in excess to ensure all acid reacts.
Effervescence in acid-base reactions indicates a carbonate is present.
Care should be taken when handling toxic salts, such as lead compounds.
Additional info: These notes cover the essential concepts and laboratory techniques for acids, bases, and salt preparations, including solubility rules, reactions, and practical methods. The step-by-step diagrams reinforce the precipitation method for preparing insoluble salts.
