 Back
BackAcids, Bases, and the pH Scale: Quantitative Aspects of Aqueous Solutions
Study Guide - Smart Notes

Acids, Bases, and pH
Defining Acids and Bases
Understanding the behavior of acids and bases in aqueous solutions is fundamental to general chemistry. Acids and bases can be defined in several ways, but the most common definitions are the Arrhenius and Brønsted-Lowry definitions.
Acid: A substance that donates a proton (H+) to another substance.
Base: A substance that accepts a proton (H+) from another substance.
These definitions help us understand how acids and bases interact in water and how they affect the concentration of hydrogen ions in solution.
The Auto-Ionization of Water
Water is unique in that it can act as both an acid and a base, a property known as amphoterism. In pure water, a small fraction of water molecules ionize to form hydronium (H3O+) and hydroxide (OH-) ions. This process is called the auto-ionization of water and is represented by the following equilibrium:
Equation:
This equilibrium is governed by the law of mass action and has an associated equilibrium constant, known as the ion-product constant for water ().
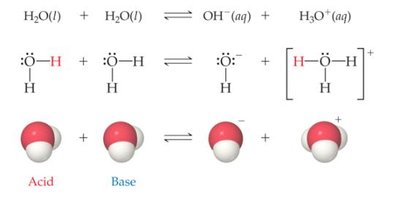
Key Points:
One water molecule acts as an acid (proton donor), and the other as a base (proton acceptor).
The products are hydroxide ion (OH-) and hydronium ion (H3O+).
Example: In pure water at 25°C, M, and .
The pH Scale and the Significance of "p"
The pH scale is a convenient way to express the acidity or basicity of a solution without using cumbersome scientific notation. The "p" in pH stands for the negative logarithm (base 10) of the hydrogen ion concentration:
pH Definition:
pOH Definition:
The sum of pH and pOH in water at 25°C is always 14:
This mathematical relationship is derived from the equilibrium constant for water ():
at 25°C
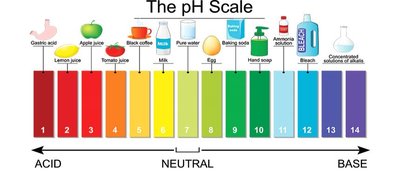
The pH scale typically ranges from 0 (very acidic) to 14 (very basic), with 7 being neutral.
Example: A solution with M has a pH of 3.
Calculating pH and pOH
To determine the acidity or basicity of a solution, you can calculate the pH or pOH using the following formulas:
pH Calculation:
pOH Calculation:
Converting between pH and [H+]:
Relationship: (at 25°C)
Example: If the pH of a solution is 5, then M. If the pOH is 3, then M, and the pH is 11.
Applications and Importance
Understanding pH is crucial in many fields, including biology, medicine, environmental science, and industry. The pH of a solution affects chemical reactivity, biological activity, and environmental processes.
Acidic solutions (pH < 7) are common in the stomach (gastric acid) and in many foods.
Basic solutions (pH > 7) are found in cleaning agents and some biological fluids.
Neutral solutions (pH = 7) include pure water.
Example: The pH of blood is tightly regulated around 7.4; deviations can be life-threatening.
