 Back
BackAcids, Bases, and the pH Scale: Quantitative Expression of Acidity and Basicity
Study Guide - Smart Notes

Acids, Bases, and pH
Defining Acids and Bases
Understanding acids and bases is fundamental to general chemistry, especially in aqueous solutions. Acids and bases are defined by their behavior in water and their ability to donate or accept protons (H+ ions).
Acid: A substance that donates a proton (H+) to another substance. In aqueous solution, acids increase the concentration of H+ ions.
Base: A substance that accepts a proton (H+) or donates a hydroxide ion (OH-). In aqueous solution, bases increase the concentration of OH- ions.
Brønsted-Lowry Definition: Acids are proton donors; bases are proton acceptors.
Arrhenius Definition: Acids produce H+ in water; bases produce OH- in water.
Example: Hydrochloric acid (HCl) is an acid because it donates H+ in water. Sodium hydroxide (NaOH) is a base because it produces OH- in water.
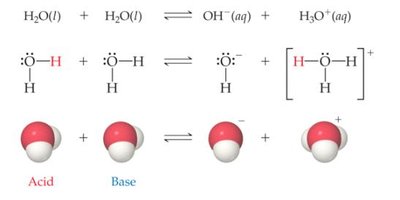
Auto-Ionization of Water
Water exhibits unique acid/base behavior due to its ability to self-ionize. This equilibrium reaction is fundamental to understanding pH and the properties of aqueous solutions.
Auto-ionization Reaction: Two water molecules react to form a hydronium ion (H3O+) and a hydroxide ion (OH-).
Equation:
This process is governed by the law of mass action and has an associated equilibrium constant, known as the ion-product constant for water ().
At 25°C:
Example: In pure water, M.
The pH Scale and Its Mathematical Basis
The pH scale is a convenient way to express the acidity or basicity of a solution, avoiding the use of scientific notation for very small concentrations. The scale is based on the equilibrium of water and the concentration of hydrogen ions.
pH Definition: pH is the negative logarithm of the hydrogen ion concentration.
Formula:
pOH: Similarly, pOH is the negative logarithm of the hydroxide ion concentration.
Formula:
Relationship: at 25°C.
Acidic Solution: pH < 7
Neutral Solution: pH = 7
Basic Solution: pH > 7
Example: If M, then (acidic).
Calculating pH and pOH
Quantitative calculations of pH and pOH are essential for expressing the acidity or basicity of solutions.
To calculate pH: Use the formula .
To calculate pOH: Use the formula .
To convert pH to [H+]:
To convert pOH to [OH-]:
Relationship:
Example: If pH = 5, then M.
Summary Table: Acidic, Neutral, and Basic Solutions
The following table summarizes the typical characteristics of acidic, neutral, and basic solutions:
Type of Solution | pH Range | [H+] | [OH-] |
|---|---|---|---|
Acidic | < 7 | > 1.0 \times 10^{-7} M | < 1.0 \times 10^{-7} M |
Neutral | 7 | 1.0 \times 10^{-7} M | 1.0 \times 10^{-7} M |
Basic | > 7 | < 1.0 \times 10^{-7} M | > 1.0 \times 10^{-7} M |
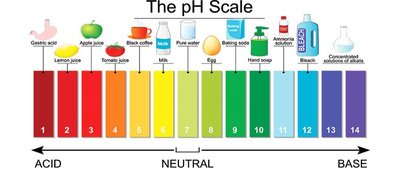
Applications and Examples
The pH scale is used in many real-world contexts, such as environmental chemistry, biology, and industrial processes. Common substances have characteristic pH values, as shown in the pH scale image above.
Acidic: Lemon juice, gastric acid
Neutral: Pure water
Basic: Bleach, ammonia solution
Example: The pH of blood is tightly regulated around 7.4, which is slightly basic.
Additional info: The concept of pH is central to understanding acid-base equilibria, buffer solutions, and titration curves in general chemistry.
