 Back
Backchem exam 4 (ch 14)-Acids, Bases, and Their Reactions: Naming, Properties, and the pH Scale
Study Guide - Smart Notes

Acids and Bases: Definitions and Properties
Arrhenius Acids and Bases
The Arrhenius definition classifies acids and bases based on their behavior in water. An Arrhenius acid is a substance that produces hydrogen ions (H+) when dissolved in water, while an Arrhenius base produces hydroxide ions (OH−).
Acids taste sour, can corrode metals, and neutralize bases.
Bases taste bitter or chalky, feel slippery, and neutralize acids.

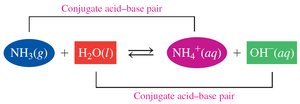
Example: Sulfuric acid (H2SO4) dissolves in water to produce H+ ions and sulfate ions (SO42−).
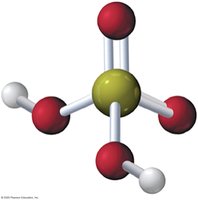
Example: Sodium hydroxide (NaOH) dissolves in water to produce Na+ and OH− ions.
Naming Acids and Bases
Naming Binary Acids
Binary acids consist of hydrogen and a nonmetal. They are named with the prefix hydro- and the suffix -ic acid.
Formula: H + nonmetal
Naming: hydro + [root of nonmetal] + ic acid
Example: HCl is hydrochloric acid; HBr is hydrobromic acid.

Naming Oxyacids
Oxyacids contain hydrogen, oxygen, and another element (usually a nonmetal). Their names are based on the polyatomic ion present:
If the ion ends in -ate, the acid name ends in -ic acid (e.g., HNO3: nitric acid).
If the ion ends in -ite, the acid name ends in -ous acid (e.g., HNO2: nitrous acid).
Prefixes per- and hypo- are used for acids with more or fewer oxygen atoms, respectively (e.g., HClO4: perchloric acid; HClO: hypochlorous acid).
Naming Bases
Most bases are named as hydroxides, consisting of a metal ion and the hydroxide ion (OH−).
Example: NaOH is sodium hydroxide; Mg(OH)2 is magnesium hydroxide.
Brønsted–Lowry Acids and Bases
Brønsted–Lowry Theory
The Brønsted–Lowry definition expands on Arrhenius by focusing on proton transfer:
Acid: Proton (H+) donor
Base: Proton (H+) acceptor
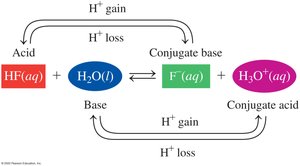
In water, free H+ ions do not exist; instead, they bond to water molecules to form the hydronium ion (H3O+).
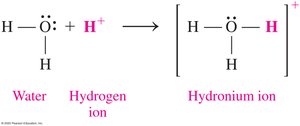
Conjugate Acid–Base Pairs
When an acid donates a proton, it forms its conjugate base. When a base accepts a proton, it forms its conjugate acid.
Example: HF (acid) + H2O (base) ⇌ F− (conjugate base) + H3O+ (conjugate acid)
The pH Scale
Definition and Calculation
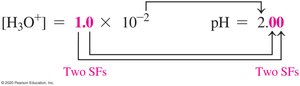
The pH scale quantifies the acidity or basicity of a solution. It is defined as the negative logarithm of the hydronium ion concentration:
pH < 7: acidic; pH = 7: neutral; pH > 7: basic (at 25°C)

Measuring pH
pH can be measured using a pH meter, pH paper, or indicators that change color at specific pH values.

Significant Figures in pH
The number of decimal places in a pH value equals the number of significant figures in the coefficient of [H3O+].
pOH and the Relationship to pH
The pOH scale is related to the hydroxide ion concentration:
At 25°C,
![Table comparing pH, pOH, [H3O+], and [OH-]](https://static.studychannel.pearsonprd.tech/study_guide_files/general-chemistry/sub_images/3b0bb4df_image_27.png)
Reactions of Acids and Bases
Acids with Metals
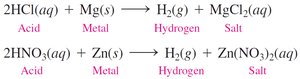
Acids react with active metals (e.g., K, Na, Ca, Mg, Al, Zn, Fe, Sn) to produce hydrogen gas and a salt. This is a single replacement reaction.
General equation:
Acids with Carbonates and Bicarbonates
Acids react with carbonates (CO32−) and bicarbonates (HCO3−) to produce carbon dioxide gas, water, and a salt.
General equation:

Neutralization Reactions
Neutralization is the reaction between an acid and a base to produce water and a salt. One H+ from the acid reacts with one OH− from the base.
General equation:

Summary Table: Common Acids, Bases, and Their Properties
Substance | Type | Name | Formula | Properties |
|---|---|---|---|---|
HCl | Acid | Hydrochloric acid | HCl | Sour, reacts with metals |
H2SO4 | Acid | Sulfuric acid | H2SO4 | Strong acid, used in industry |
NaOH | Base | Sodium hydroxide | NaOH | Slippery, caustic |
NH3 | Base | Ammonia | NH3 | Pungent, cleaning agent |
Key Equations
(at 25°C)
Review and Applications
Acids and bases are essential in biological, environmental, and industrial processes.
Understanding naming conventions, reactions, and the pH scale is fundamental for predicting chemical behavior and for laboratory work.
