Back
BackAdditional Aspects of Aqueous Equilibria: Buffers, Titrations, and Solubility
Study Guide - Smart Notes

Aqueous Equilibria
Effect of a Common Ion on Acid-Base Equilibria
When a weak acid is in solution with a salt containing its conjugate base, the equilibrium shifts according to Le Châtelier’s Principle. The presence of a common ion suppresses the ionization of the weak acid or base.
Example: Acetic acid and sodium acetate in solution:
CH3COOH(aq) ↔ H+(aq) + CH3COO−(aq)
NaCH3COO(aq) → Na+(aq) + CH3COO−(aq)
The addition of acetate ion shifts the equilibrium to the left, decreasing [H+].

The Common-Ion Effect
The common-ion effect describes the decrease in the degree of dissociation of a weak electrolyte when a strong electrolyte containing a common ion is added to the solution. This principle is crucial in acid-base equilibria and solubility equilibria.
Key Point: The weak electrolyte ionizes less in the presence of a common ion.
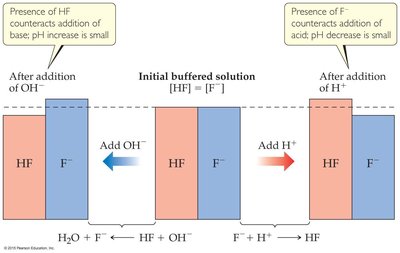
Buffer Solutions
Buffers are solutions that resist drastic changes in pH when small amounts of acid or base are added. They consist of a weak acid and its conjugate base or a weak base and its conjugate acid, both at relatively high concentrations.
Preparation: Mix a weak acid with a salt of its conjugate base, or a weak base with a salt of its conjugate acid.
Alternatively, partially neutralize a weak acid with a strong base or a weak base with a strong acid.

How Buffers Work
Buffers function by neutralizing added acids (H+) or bases (OH−), maintaining a relatively constant pH. The weak acid neutralizes added base, while the conjugate base neutralizes added acid.
Calculating the pH of a Buffer: The Henderson–Hasselbalch Equation
The Henderson–Hasselbalch equation provides a convenient way to calculate the pH of a buffer solution:
Where [A−] is the concentration of the conjugate base and [HA] is the concentration of the weak acid.
This equation is valid only for buffer solutions.
Buffer Capacity and pH Range
Buffer capacity is the amount of acid or base a buffer can neutralize before the pH changes significantly. The effective pH range of a buffer is typically within ±1 pH unit of the pKa of the acid.
Optimal Buffering: When [HA] = [A−], pH = pKa.
Buffer Calculations with Added Strong Acid or Base
When a strong acid or base is added to a buffer, a stoichiometric calculation is performed to determine the new concentrations of the acid and base forms. The Henderson–Hasselbalch equation is then used to find the new pH.
Titrations
Principles of Titration
Titration is a technique in which a solution of known concentration (titrant) is added to a solution of unknown concentration until the reaction reaches the equivalence point, where stoichiometrically equivalent amounts of acid and base have reacted.
Titration Curves
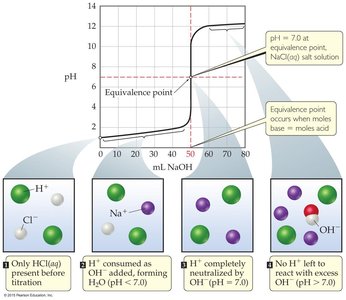
Strong Acid with Strong Base: The pH rises slowly at first, then rapidly near the equivalence point (pH = 7), and then levels off.
Strong Base with Strong Acid: The curve is the mirror image, starting at high pH and ending at low pH, with equivalence at pH = 7.
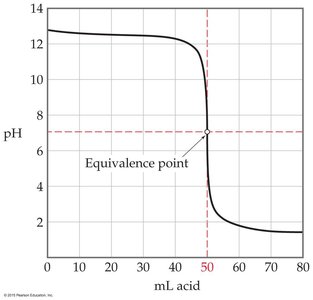
Weak Acid with Strong Base: The initial pH is higher, there is a buffer region, and the equivalence point is above pH 7 due to the formation of a weak conjugate base.
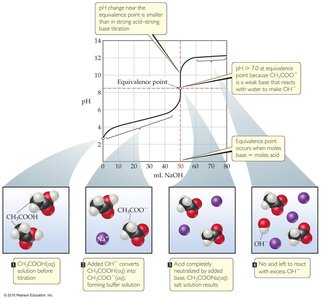
Comparison: The pH change near the equivalence point is smaller for weak acids, and the equivalence point pH is greater than 7.
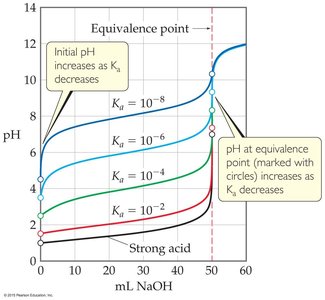
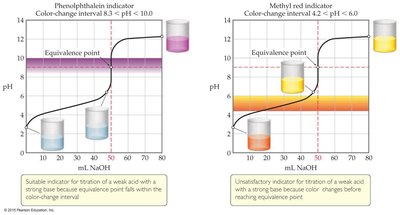
Indicators in Titration
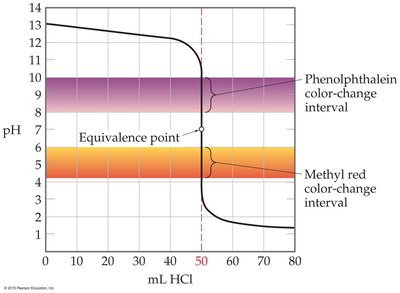
Indicators are weak acids or bases that change color over a specific pH range. The choice of indicator is critical and should match the pH at the equivalence point of the titration.
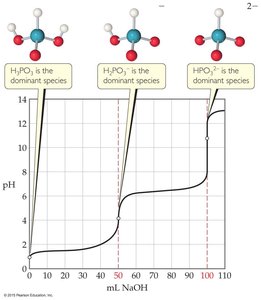
Titration of Polyprotic Acids
Polyprotic acids have more than one ionizable proton and thus exhibit multiple equivalence points during titration. Each equivalence point corresponds to the neutralization of one proton.
Solubility Equilibria
Solubility Product Constant (Ksp)
The solubility product constant, Ksp, describes the equilibrium between a solid ionic compound and its dissolved ions in a saturated solution.
Example: For BaSO4(s):
For Ba3(PO4)2(s):
Solubility vs. Solubility Product
Solubility is the amount of solute that dissolves to form a saturated solution, usually expressed in g/L or mol/L. Ksp is the equilibrium constant for the dissolution process.
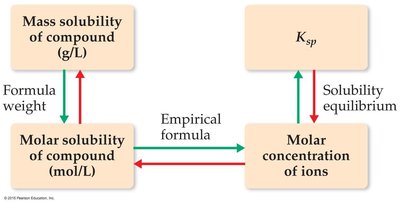
Calculating Solubility from Ksp
To find the molar solubility from Ksp, set up an ICE table and solve for the concentration of ions at equilibrium.
Example: For CaF2,
Let x = molar solubility, then ,
So,
Factors Affecting Solubility
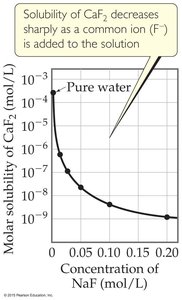
Common-Ion Effect: The presence of a common ion decreases the solubility of a salt.
pH: Salts with basic anions are more soluble in acidic solutions.
Complex Ion Formation: Metal ions can form complex ions with Lewis bases, increasing solubility.
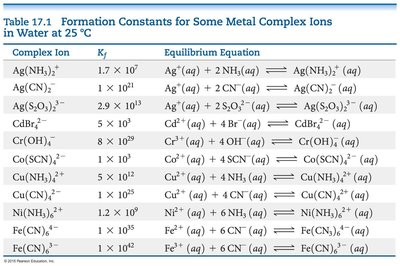
Complex Ion Formation
Complex ions are formed when metal ions bind to ligands (Lewis bases). The formation of complex ions can greatly increase the solubility of otherwise insoluble salts.
Complex Ion | Kf | Equilibrium Equation |
|---|---|---|
Ag(NH3)2+ | 1.7 × 107 | Ag+(aq) + 2 NH3(aq) ↔ Ag(NH3)2+(aq) |
Ag(CN)2− | 1 × 1021 | Ag+(aq) + 2 CN−(aq) ↔ Ag(CN)2−(aq) |
Ag(S2O3)23− | 2.9 × 1013 | Ag+(aq) + 2 S2O32−(aq) ↔ Ag(S2O3)23−(aq) |
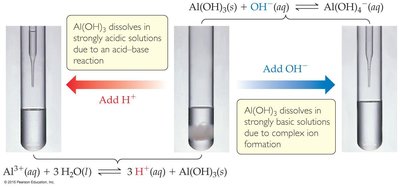
Amphoterism and Solubility
Amphoteric oxides and hydroxides can dissolve in both strong acids and bases due to their ability to act as either acids or bases. Examples include Al(OH)3, Cr(OH)3, Zn(OH)2, and Sn(OH)2.
Predicting Precipitation: The Reaction Quotient (Q)
To determine if a precipitate will form, compare the reaction quotient (Q) to Ksp:
If Q = Ksp, the solution is saturated (at equilibrium).
If Q < Ksp, more solid can dissolve (no precipitate forms).
If Q > Ksp, a precipitate will form.
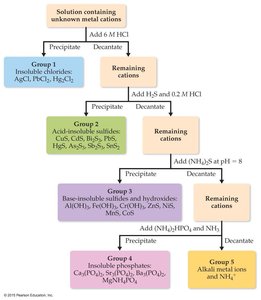
Selective Precipitation of Ions
Selective precipitation uses differences in solubility to separate ions in a mixture. This technique is fundamental in qualitative analysis for identifying ions in solution.