 Back
Backchapter 18
Study Guide - Smart Notes

Amines
Definition and Structure
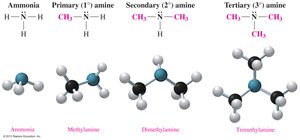
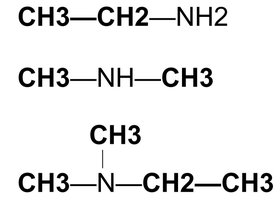
Amines are organic compounds derived from ammonia (NH3) by replacing one or more hydrogen atoms with alkyl or aryl groups. They are classified as primary (1°), secondary (2°), or tertiary (3°) depending on the number of carbon-containing groups attached to the nitrogen atom.
Primary amine (1°): One carbon group attached to nitrogen.
Secondary amine (2°): Two carbon groups attached to nitrogen.
Tertiary amine (3°): Three carbon groups attached to nitrogen.
Nomenclature of Amines
The IUPAC naming of amines involves replacing the "-e" ending of the parent alkane with "-amine." For chains of three or more carbons, the position of the –NH2 group is indicated by a number. Alkyl groups attached to the nitrogen are indicated by the prefix "N-" and listed alphabetically.
Simple amines: Methanamine (from methane), Ethanamine (from ethane).
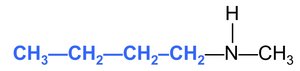
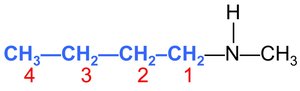
Substituted amines: N-methyl-1-butanamine (a methyl group attached to the nitrogen of butanamine).
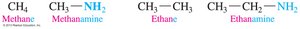
Common Names of Amines
Common names are formed by listing the alkyl groups attached to the nitrogen in alphabetical order, followed by "amine." For example, ethylamine, dimethylamine, and ethyldimethylamine.
Naming Amines with Multiple Functional Groups
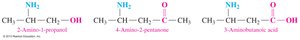
When a molecule contains more than one functional group, the group with the highest priority determines the suffix, while others are named as substituents. The amino group (–NH2) is named as "amino" when not the highest priority.
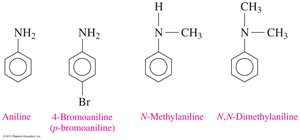
Aromatic Amines
Aromatic amines are derivatives of aniline (aminobenzene). Substituents on the nitrogen are indicated with the prefix "N-" and the alkyl name.
Classification of Amines
Amines are classified based on the number of carbon groups attached to the nitrogen atom:
Primary (1°): RNH2
Secondary (2°): R2NH
Tertiary (3°): R3N

Line-Angle Formulas for Amines
In line-angle formulas, the hydrogen atom bonded to nitrogen must be explicitly shown to distinguish between primary, secondary, and tertiary amines.
Properties of Amines
Hydrogen Bonding and Boiling Points
Amines contain polar N–H bonds, allowing primary and secondary amines to form hydrogen bonds with each other. However, these bonds are weaker than those in alcohols due to the lower electronegativity of nitrogen compared to oxygen. Tertiary amines cannot form hydrogen bonds with each other.
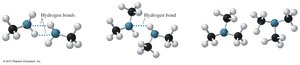
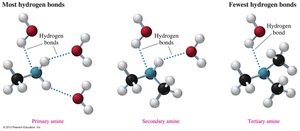
Boiling points: Primary > Secondary > Tertiary (for similar molar mass).
Solubility in Water
Amines with 1 to 6 carbon atoms are generally soluble in water due to their ability to form hydrogen bonds with water molecules. Solubility decreases as the size of the alkyl group increases.
Basicity of Amines
Amines act as Brønsted-Lowry bases, accepting a proton (H+) from water to form an ammonium ion and a hydroxide ion. They are considered weak bases in aqueous solution.
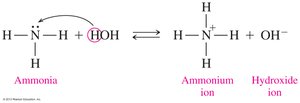
Formation of Ammonium Salts
When an amine reacts with an acid, it forms an ammonium salt, which is ionic, solid at room temperature, and highly soluble in water. Quaternary ammonium salts have four carbon groups attached to the nitrogen and are important in biological systems (e.g., choline).
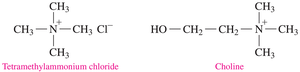
Heterocyclic Amines and Alkaloids
Heterocyclic Amines
Heterocyclic amines are cyclic compounds containing nitrogen atoms in the ring. They are found in many biologically important molecules, such as DNA and RNA bases, and in natural products like piperidine (in black pepper).

Alkaloids
Alkaloids are physiologically active nitrogen-containing compounds produced by plants. Examples include nicotine, caffeine, morphine, and codeine. Many alkaloids are used as stimulants, anesthetics, or painkillers.

Neurotransmitters
Definition and Function
Neurotransmitters are chemical compounds that transmit electrical impulses from nerve cells to target cells (other nerves, muscles, or glands). Many neurotransmitters are amines or contain amine groups.
Examples of Amine Neurotransmitters
Acetylcholine: Involved in muscle contraction and memory. Low levels are associated with Alzheimer's disease.
Catecholamines: Dopamine, norepinephrine, and epinephrine are derived from tyrosine and regulate mood, alertness, and the fight-or-flight response.
Serotonin: Synthesized from tryptophan, it regulates mood, sleep, and well-being. Low levels are linked to depression and anxiety.
Histamine: Involved in immune response and allergic reactions.
Glutamate and GABA: Glutamate is the most abundant excitatory neurotransmitter, while GABA is the most common inhibitory neurotransmitter in the brain.
Amides
Structure and Nomenclature
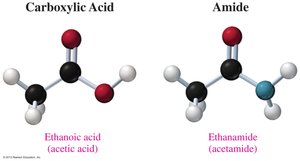
Amides are derivatives of carboxylic acids in which the –OH group is replaced by an amino group (–NH2, –NHR, or –NR2). They are named by replacing the "-ic acid" or "-oic acid" ending of the parent acid with "amide." Substituents on the nitrogen are indicated with "N-" prefixes.

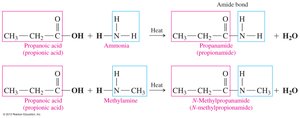
Preparation of Amides (Amidation)
Amides are prepared by heating a carboxylic acid with ammonia or a primary/secondary amine. This reaction is called amidation and produces an amide and water.
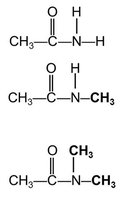
Classification of Amides
Amides are classified as primary, secondary, or tertiary based on the number of carbon groups attached to the nitrogen atom.
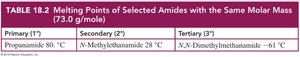
Properties of Amides
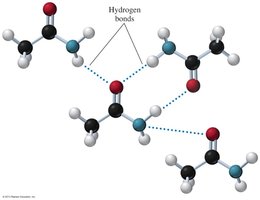
Melting Points: Primary and secondary amides can form hydrogen bonds, resulting in higher melting points. Tertiary amides, lacking N–H bonds, have lower melting points.
Solubility: Amides with 1 to 5 carbon atoms are soluble in water due to hydrogen bonding with water molecules.
Biological and Medical Importance
Amides are found in many biologically important molecules, including proteins (peptide bonds), urea (waste product of protein metabolism), and pharmaceuticals such as acetaminophen and barbiturates.
Hydrolysis of Amides
Amides can be hydrolyzed (broken down by water) in the presence of acid or base. Acid hydrolysis produces a carboxylic acid and an ammonium salt, while base hydrolysis produces the salt of the carboxylic acid and an amine or ammonia.
Acid hydrolysis:
Base hydrolysis:
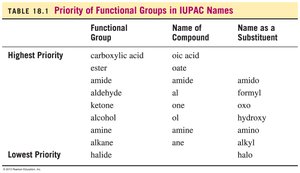
Summary Table: Priority of Functional Groups in IUPAC Names
Functional Group | Name of Compound | Name as a Substituent |
|---|---|---|
Carboxylic acid | oic acid | carboxy |
Ester | oate | alkoxycarbonyl |
Amide | amide | amido |
Aldehyde | al | formyl |
Ketone | one | oxo |
Alcohol | ol | hydroxy |
Amine | amine | amino |
Alkene | ene | alkenyl |
Alkyne | yne | alkynyl |
Alkane | ane | alkyl |
Halide | halo | halo |
