 Back
BackAnalyzing a Titration Curve: Diprotic Acid with Strong Base
Study Guide - Smart Notes

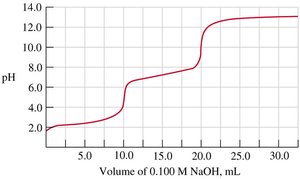
Q8. The following titration curve indicates the titration of what kinds of substances?
Background
Topic: Acid-Base Titration Curves
This question tests your ability to interpret titration curves and identify the types of acids and bases involved based on the shape and features of the curve.
Key Terms and Concepts:
Titration Curve: A plot of pH versus volume of titrant added, used to analyze acid-base reactions.
Equivalence Point: The point at which stoichiometrically equivalent amounts of acid and base have reacted.
Diprotic Acid: An acid that can donate two protons (H+).
Monoprotic Acid: An acid that can donate only one proton.
Strong Base: A base that completely dissociates in water (e.g., NaOH).
Step-by-Step Guidance
Examine the titration curve for the number of distinct inflection points or "steps." Each step typically corresponds to an equivalence point for a proton being neutralized.
Notice that the curve has two distinct jumps in pH, indicating two equivalence points. This suggests the acid being titrated can donate two protons (i.e., it is diprotic).
Observe the starting pH (around 2), which is typical for a weak acid, and the final pH (around 12–14), which is typical for titration with a strong base.
Compare the curve to typical titration curves for monoprotic and diprotic acids. Monoprotic acids have one equivalence point, while diprotic acids have two.
Try solving on your own before revealing the answer!
Final Answer: C) diprotic acid with a strong base
The two distinct jumps in the titration curve indicate two equivalence points, characteristic of a diprotic acid being titrated with a strong base.
