 Back
BackAqueous Ionic Equilibria: Buffers and Acid-Base Titrations
Study Guide - Smart Notes

Buffer Solutions
Definition, Components, and Properties
Buffer solutions are mixtures that resist changes in pH when small amounts of strong acid or base are added. They consist of a weak acid and its conjugate base, or a weak base and its conjugate acid.
Components: Typically, a weak acid (HA) and its salt (A−), or a weak base (B) and its salt (BH+).
Properties: Buffers maintain pH stability, crucial in chemical and biological systems.
Example: Acetic acid (HC2H3O2) and sodium acetate (NaC2H3O2).
Understanding Buffer Action
Buffer action relies on the equilibrium between the weak acid and its conjugate base. When strong acid is added, the base component neutralizes it; when strong base is added, the acid component neutralizes it.
Le Chatelier's Principle: Addition of acid or base shifts the equilibrium, minimizing pH change.
Henderson-Hasselbalch Equation and pH Calculation
Equation and Application
The Henderson-Hasselbalch equation relates the pH of a buffer to the concentrations of its acid and base components:
Equation:
Application: Used to calculate buffer pH and to design buffers with desired pH values.
Preparation of Buffer Solutions
From Available Chemicals and Solutions
To prepare a buffer, select a weak acid/base pair with a pKa close to the desired pH. Adjust the ratio of acid to base using the Henderson-Hasselbalch equation.
Example: Preparing 500 mL of pH 4.25 buffer from 1.00 M acetic acid and solid sodium acetate:
Calculate the required ratio using the equation.
Determine the mass of sodium acetate needed:
Dissolve in acetic acid, dilute to volume, and adjust pH as needed.
Buffer Capacity and Range
Definition and Quantitative Description
Buffer capacity measures a buffer's ability to resist pH changes. It is highest when the concentrations of acid and base are equal.
Quantitative: Number of moles of strong acid/base causing a 1 unit pH change in 1 L of buffer.
Buffer Range: Effective within pKa ± 1.
Best Buffer: When [base]/[acid] ≈ 1, pH ≈ pKa.
Buffer Capacity Example
Two buffers with identical pH but different concentrations have different capacities. Higher concentrations yield greater capacity.
Calculations Involving Buffers Using the ICE Table
ICE Table Method
The ICE (Initial, Change, Equilibrium) table is used to calculate equilibrium concentrations and pH in buffer solutions.
Example: 0.30 M acetic acid and 0.20 M sodium acetate buffer.
Set up the equilibrium:
Calculate pH using both ICE table and Henderson-Hasselbalch equation.
Common Ion Effect
The presence of a common ion suppresses ionization, reducing % ionization compared to a solution containing only the weak acid.
Example: % ionization in buffer is much lower than in pure acetic acid solution.
Buffer Response to Strong Acid/Base Addition
Strong base (NaOH) reacts with acid component; strong acid (HCl) reacts with base component.
Use stoichiometry to update concentrations, then recalculate pH.
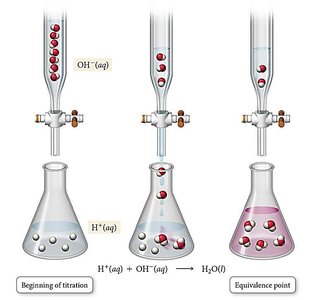
Acid-Base Titrations
Terminology and Concepts
Acid-base titrations determine the concentration of an unknown acid or base by reacting it with a titrant of known concentration.
Titrant: Solution added from a buret.
Analyte: Solution being analyzed.
Equivalence Point: Stoichiometric amount of titrant added.
Endpoint: Detected by indicator color change; ideally coincides with equivalence point.
Titration Error: Difference between endpoint and equivalence point.
Acid-Base Titration Curves
During titration, pH changes as titrant is added. A pH titration curve plots pH versus volume of titrant.
Information from Curve: Equivalence point, unknown concentration, dissociation constants (Ka, Kb), and molecular weight.
Categories: Strong acid-strong base, weak acid-strong base, weak base-strong acid, polyprotic acid-strong base.

Strong Acid - Strong Base Titration Curve Example
Consider titrating 50.00 mL of 0.10 M HCl with 0.10 M NaOH. The pH is calculated at various stages:
Volume NaOH Added (mL) | Region | Description | Anticipated pH |
|---|---|---|---|
0.00 | Initial solution | Strong acid solution | Acidic (pH = 1.00) |
25.00 | Halfway point | Half of strong acid consumed | Acidic (pH = 1.48) |
50.00 | Equivalence point | All strong acid consumed | Neutral (pH = 7.00) |
55.00 | Post-equivalence point | Excess base solution | Basic (pH = 11.68) |
Equivalence Point: Calculated by stoichiometry; at this point, all acid is neutralized.
Post-Equivalence: Excess base determines pH.
Key Equations:
pH calculation:
pOH calculation:
Relationship:
Example Calculations:
At equivalence, from water autoionization:
Post-equivalence, excess determines pH.
Additional info: Laboratory titrations often use pH meters and magnetic stirrers for accurate measurement and mixing.
