 Back
BackAqueous Ionic Equilibrium: Buffers, Titrations, and Solubility
Study Guide - Smart Notes

Chapter 18: Aqueous Ionic Equilibrium
Buffer Solutions
Buffer solutions are essential in maintaining a relatively constant pH when small amounts of acid or base are added. They are widely used in chemical, biological, and industrial processes where pH stability is crucial.
Definition: A buffer is a solution that resists changes in pH upon the addition of small amounts of acid or base.
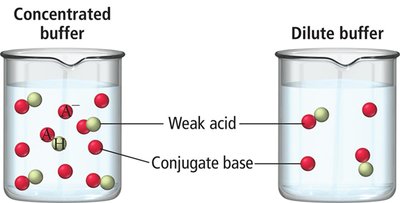
Composition: Buffers typically consist of significant amounts of a weak acid and its conjugate base, or a weak base and its conjugate acid.
Mechanism: The weak acid neutralizes added base, while the conjugate base neutralizes added acid, keeping the pH nearly constant.
Concentration: Effective buffers contain about 10-3 M of both the acid and base pair.
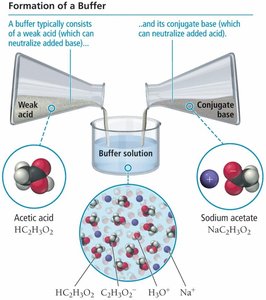
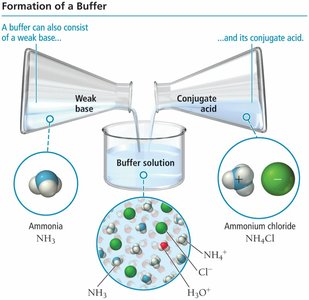
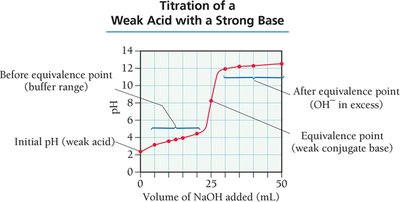
Acidic and Basic Buffer Solutions
Acidic buffers contain a weak acid and its conjugate base, while basic buffers contain a weak base and its conjugate acid. The buffer's action is explained by Le Châtelier’s principle, which describes how equilibrium shifts to counteract changes in concentration.
Acidic Buffer Example: Acetic acid (HC2H3O2) and sodium acetate (NaC2H3O2).
Basic Buffer Example: Ammonia (NH3) and ammonium chloride (NH4Cl).
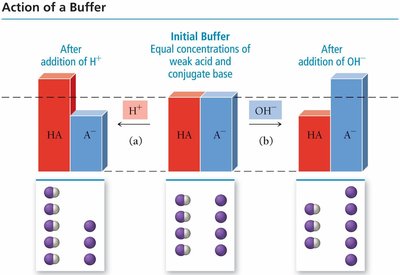
How Buffers Work
Buffers maintain pH by neutralizing added acids or bases. The weak acid component reacts with added base, and the conjugate base component reacts with added acid. This process keeps the concentration of H3O+ ions nearly constant.
Addition of Base: HA + OH- → A- + H2O
Addition of Acid: A- + H+ → HA
Common-Ion Effect
The common-ion effect occurs when a solution contains two substances that share a common ion. The presence of the common ion suppresses the ionization of the weak electrolyte, shifting the equilibrium and affecting pH.
Example: Adding sodium acetate to acetic acid solution decreases the ionization of acetic acid, lowering [H3O+] and increasing pH.
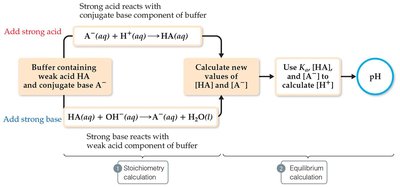
Calculating the pH of Buffer Solutions
To determine the pH of a buffer, use the equilibrium (ICE) table or the Henderson-Hasselbalch equation. The ICE table approach involves setting up initial concentrations, changes, and equilibrium concentrations, then solving for [H3O+].
Henderson-Hasselbalch Equation:
Use this equation when the initial concentrations of acid and base are much larger than Ka.
Buffer Capacity and Range
Buffer capacity is the amount of acid or base a buffer can neutralize before the pH changes significantly. Buffer range is the pH range over which the buffer is effective, typically within ±1 pH unit of the buffer's pKa.
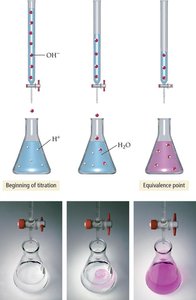
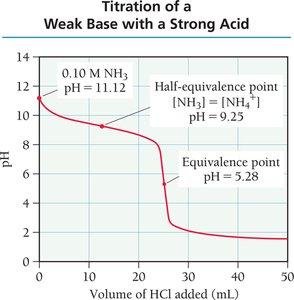
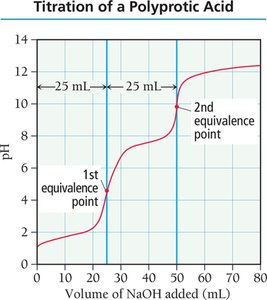
Titration and Titration Curves
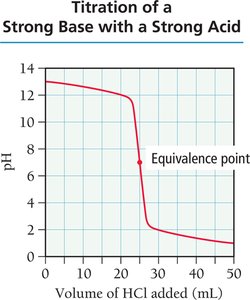
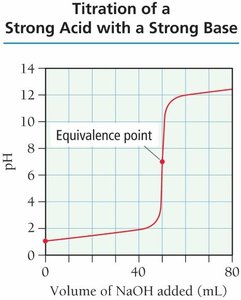
Titration is a technique used to determine the concentration of an unknown solution by reacting it with a solution of known concentration. The titration curve plots pH versus the volume of titrant added, revealing important points such as the equivalence point.
Equivalence Point: The point at which stoichiometrically equivalent amounts of acid and base have reacted.
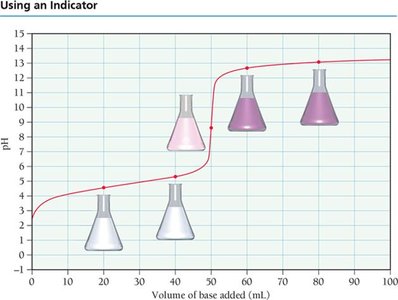
Indicators: Chemicals that change color at specific pH values, used to detect the endpoint of titration.
Indicators and Their Color Changes
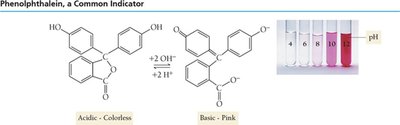
Indicators are weak acids or bases that exhibit different colors in their acid and base forms. The color change occurs over a specific pH range, which is related to the indicator's pKa.
Phenolphthalein: Colorless in acid, pink in base.
Methyl Red: Red in acid, yellow in base.
pH (relative to pKa) | [In-]/[HIn] Ratio | Color of Indicator Solution |
|---|---|---|
pH = pKa | 1 | Intermediate color |
pH = pKa + 1 | 10 | Color of In- |
pH = pKa - 1 | 0.1 | Color of HIn |


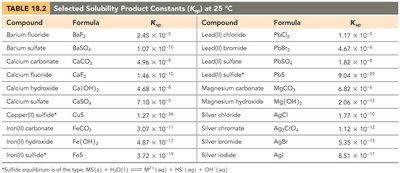
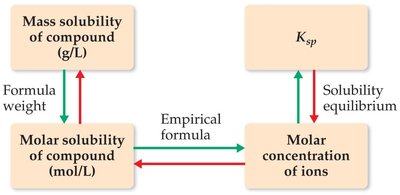
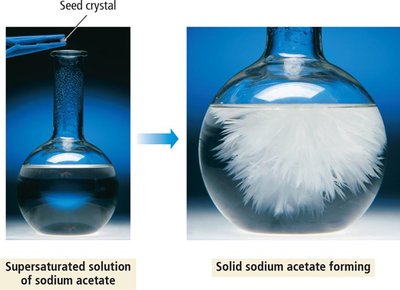
Solubility-Product Constant (Ksp)
The solubility-product constant, Ksp, describes the equilibrium between a solid ionic compound and its dissolved ions in a saturated solution. It is used to predict the solubility of sparingly soluble salts and to determine whether precipitation will occur.
Solubility: The amount of solute that dissolves to form a saturated solution (g/L or mol/L).
Ksp Expression: For BaSO4:
Common-Ion Effect on Solubility
The addition of a soluble salt containing a common ion decreases the solubility of an insoluble salt by shifting the equilibrium to the left. This is an application of Le Châtelier’s principle.
Effect of pH on Solubility
The solubility of ionic compounds containing basic anions increases as pH decreases (solution becomes more acidic), because the added H3O+ reacts with the basic anion, removing it from solution and shifting the equilibrium toward more dissolution.
Precipitation and the Reaction Quotient (Q)
Precipitation occurs when the product of the ion concentrations exceeds Ksp. The reaction quotient, Q, is compared to Ksp to predict precipitation:
Q = Ksp: Solution is saturated, no precipitation.
Q < Ksp: Solution is unsaturated, no precipitation.
Q > Ksp: Precipitation occurs until Q = Ksp.

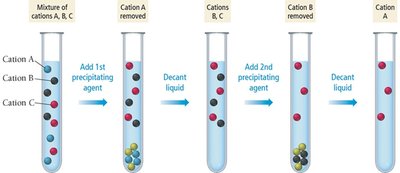
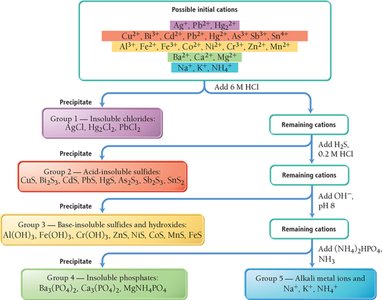
Selective Precipitation and Qualitative Analysis
Selective precipitation is used to separate ions in a mixture by adding a reagent that forms an insoluble salt with one ion but not others. This technique is fundamental in qualitative analysis schemes for identifying ions in solution.

Complex Ion Formation
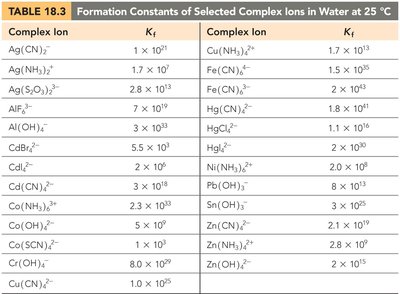
Transition metals can form complex ions with ligands, which are molecules or ions that donate electron pairs. The formation of complex ions can increase the solubility of certain salts by shifting the equilibrium.
Formation Constant (Kf): The equilibrium constant for the formation of a complex ion.
Example:
Solubility of Amphoteric Metal Hydroxides
Amphoteric hydroxides, such as Al(OH)3, can dissolve in both acidic and basic solutions. In acidic solutions, they react with H3O+ to form soluble metal ions; in basic solutions, they react with OH- to form soluble complex ions.
Example: Al(OH)3(s) + OH-(aq) → [Al(OH)4]-(aq)
Additional info: These notes cover the core concepts of aqueous ionic equilibrium, including buffer systems, titration analysis, solubility equilibria, and qualitative analysis, as relevant to a general chemistry curriculum.
