 Back
BackAqueous Ionic Equilibrium: Buffers, Titrations, and Solubility
Study Guide - Smart Notes

Aqueous Ionic Equilibrium
Buffered Solutions
Buffered solutions are essential in chemistry because they resist changes in pH when small amounts of strong acids or bases are added. Buffers are typically composed of a weak acid and its conjugate base, or a weak base and its conjugate acid.
Definition: A buffer is a solution that minimizes pH changes upon the addition of acid or base.
Composition: Buffers contain significant amounts of both a weak acid and its conjugate base (or weak base and its conjugate acid).
Mechanism: Added H+ ions react with the conjugate base, while added OH− ions react with the weak acid.
Buffering Capacity: The amount of acid or base a buffer can neutralize before the pH changes significantly.

Example: A buffer made from 0.50 M acetic acid (CH3COOH) and 0.50 M sodium acetate (CH3COONa) has a pH of 4.74. Adding 0.010 mol NaOH only raises the pH to 4.76, demonstrating the buffer's resistance to pH change.
Calculating the pH of a Buffer Solution
The pH of a buffer can be calculated using equilibrium tables or the Henderson-Hasselbalch equation. The equilibrium approach involves setting up an ICE (Initial, Change, Equilibrium) table for the weak acid dissociation.
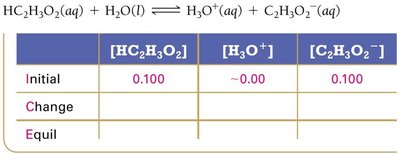
Henderson-Hasselbalch Equation:
$\mathrm{pH} = \mathrm{p}K_a + \log \left( \frac{[\text{base}]}{[\text{acid}]} \right)$
This equation is especially useful when the concentrations of the acid and its conjugate base are known.
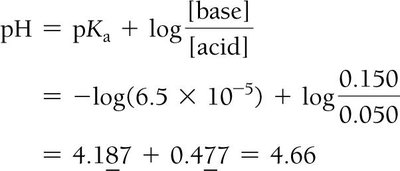
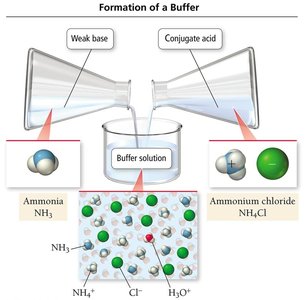
Basic Buffers
Basic buffers are prepared by mixing a weak base with a soluble salt of its conjugate acid. For example, a buffer can be made from ammonia (NH3) and ammonium chloride (NH4Cl).
Reaction: NH3(aq) + H2O(l) → NH4+(aq) + OH−(aq)
Buffering Action: Added acid reacts with NH3; added base reacts with NH4+.
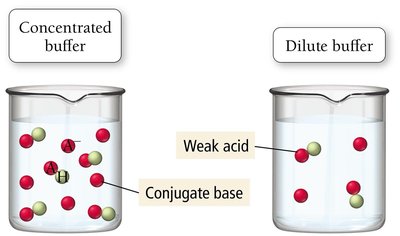
Buffering Capacity and Range
The effectiveness of a buffer depends on the absolute and relative concentrations of the acid and base components.
Buffering Capacity: Increases with higher concentrations of buffer components.
Buffering Range: Most effective when the ratio [base]:[acid] is between 0.1 and 10. The effective pH range is pKa ± 1.
Titrations and Titration Curves
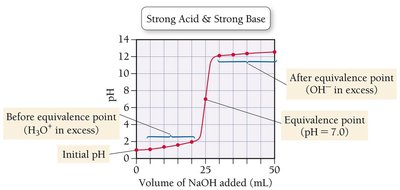
Strong Acid–Strong Base Titration
In a titration, a solution of known concentration (titrant) is added to a solution of unknown concentration until the reaction reaches the equivalence point. For strong acid–strong base titrations, the pH changes rapidly near the equivalence point.
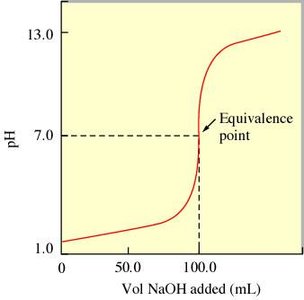
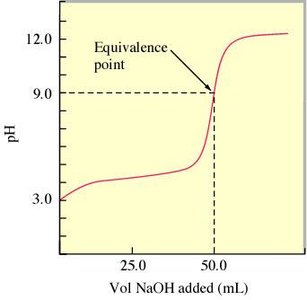
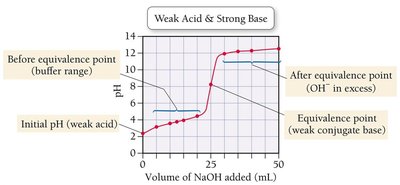
Weak Acid–Strong Base Titration
When titrating a weak acid with a strong base, the titration curve differs from that of a strong acid. The initial pH is higher, and the equivalence point pH is above 7 due to the formation of a weak conjugate base.
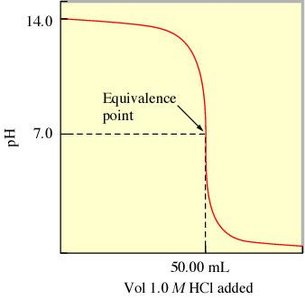
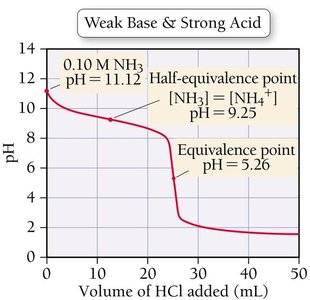
Strong Acid–Weak Base Titration
In this titration, the initial pH is high, and the equivalence point pH is below 7 due to the formation of a weak conjugate acid.
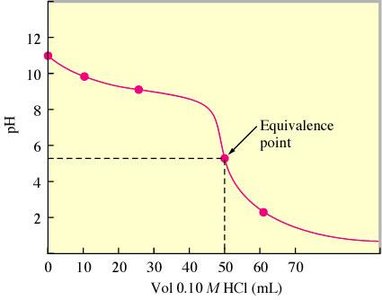
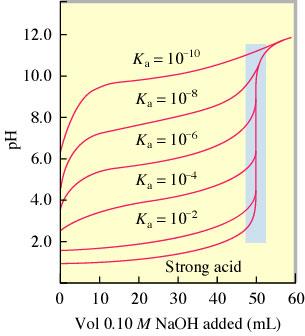
Comparing Titration Curves
Titration curves can be compared to understand the differences between strong and weak acids and bases.
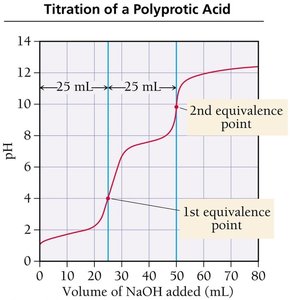
Titration of Polyprotic Acids
Polyprotic acids have more than one ionizable proton and thus show multiple equivalence points in their titration curves. The separation between equivalence points depends on the difference between their Ka values.
Acid-Base Indicators
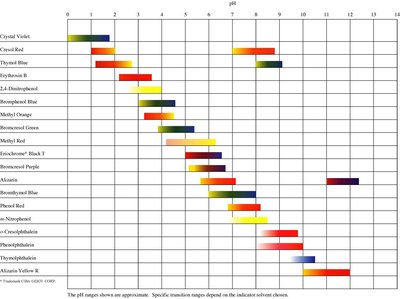
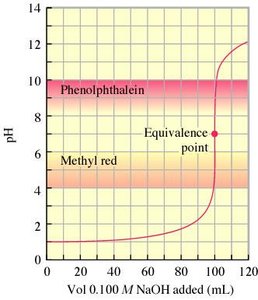
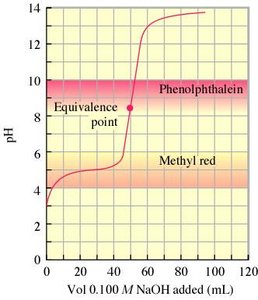
Indicators and End Points
Acid-base indicators are weak acids or bases that change color at specific pH ranges. They are used to mark the end point of a titration, which should be as close as possible to the equivalence point.



Selection of Indicator: Choose an indicator with a pKa near the equivalence point pH.



Solubility Equilibria
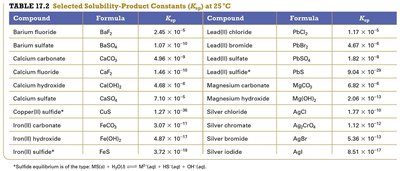
Solubility-Product Constant (Ksp)
The solubility-product constant, Ksp, is the equilibrium constant for the dissolution of a sparingly soluble ionic compound in water.
General Form: For MnXm(s) ↔ nMm+(aq) + mXn−(aq),
$K_{sp} = [M^{m+}]^n [X^{n-}]^m$
Example: For PbCl2(s) ↔ Pb2+(aq) + 2Cl−(aq),
$K_{sp} = [Pb^{2+}][Cl^-]^2$
Common Ion Effect
The solubility of an ionic compound decreases in the presence of a common ion due to Le Châtelier's principle. For example, the solubility of Ag2CrO4 is lower in a solution already containing Ag+ ions.
Predicting Precipitation
Precipitation occurs when the ionic product (Q) exceeds Ksp. If Q > Ksp, precipitation occurs; if Q < Ksp, no precipitation occurs; if Q = Ksp, the solution is saturated.


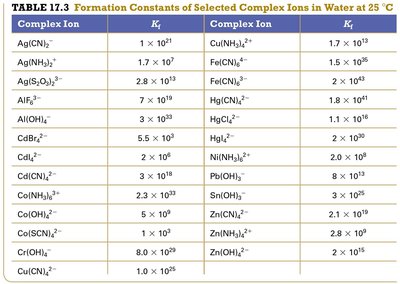
Complex Ion Formation
Transition metals can form complex ions with ligands, which increases their solubility. The equilibrium constant for complex ion formation is called the formation constant (Kf).
Solubility of Amphoteric Hydroxides
Some metal hydroxides are amphoteric, meaning they can dissolve in both acidic and basic solutions. For example, Al(OH)3 is more soluble in both acidic and basic conditions due to its ability to react as either an acid or a base.
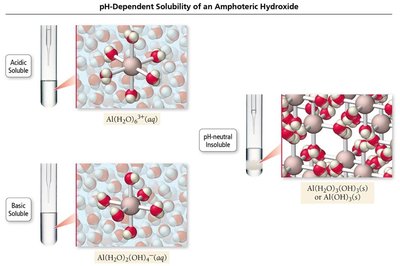
Additional info: Amphoteric substances can act as both acids and bases. Water is a classic example of an amphoteric molecule.
