 Back
BackAtomic Structure, Chemical Bonding, and Acids/Bases: Study Guide
Study Guide - Smart Notes

Atomic Structure and Properties
Atomic Number and Mass Number
The atomic structure of an element is defined by its number of protons, neutrons, and electrons. The atomic number is the number of protons in the nucleus and uniquely identifies the element. The mass number is the sum of protons and neutrons.
Atomic Number: For sodium, the atomic number is 11 (11 protons).
Mass Number: For sodium with 11 protons and 12 neutrons, the mass number is .
Example: Sodium (Na) is identified by atomic number 11 and mass number 23.
Chemical Bonding
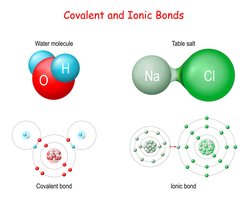
Ionic vs. Covalent Bonding
Chemical bonds are formed to achieve stable electron configurations. The two main types are ionic bonds and covalent bonds.
Ionic Bonds: Formed when one atom transfers electrons to another, resulting in charged ions. Typically occurs between metals and non-metals.
Covalent Bonds: Formed when atoms share electron pairs to fill their valence shells. Usually occurs between two non-metals.
Example: Sodium chloride (NaCl) is an ionic compound; water (H2O) is a covalent compound.
The Stability of Noble Gases
Noble gases (Group 18) are chemically stable because they have full valence electron shells. This makes them largely unreactive and unlikely to form bonds.
Key Point: Noble gases have 8 valence electrons (except Helium, which has 2).
Example: Argon, Neon, and Helium are noble gases that rarely participate in chemical reactions.
Chemical Reactions and Quantities
Balancing Chemical Equations
Balancing chemical equations ensures the Law of Conservation of Mass is followed: atoms are neither created nor destroyed in a reaction.
Unbalanced Equation:
Balanced Equation:
Explanation: Balancing requires equal numbers of each atom on both sides. Here, 4 hydrogen atoms and 2 oxygen atoms are present in both reactants and products.
Acids, Bases, and the pH Scale
Identifying Acids and Bases
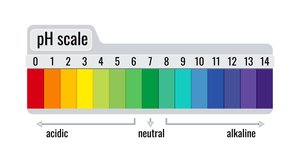
The pH scale measures the concentration of hydrogen ions in a solution, indicating whether it is acidic, neutral, or basic.
Acidic: pH 0–6 (red/orange on indicator strips); strong acids have low pH (e.g., pH 2).
Neutral: pH 7 (green).
Basic (Alkaline): pH 8–14 (blue/purple).
Example: A substance with pH 2 is a strong acid; pH 12 is a strong base.
Summary Table: Types of Chemical Bonds
Bond Type | Electron Behavior | Typical Elements | Example Compound |
|---|---|---|---|
Ionic | Transfer of electrons | Metal + Non-metal | NaCl (table salt) |
Covalent | Sharing of electrons | Non-metal + Non-metal | H2O (water) |
