 Back
BackAtomic Structure, Light, and Quantum Theory: Study Notes
Study Guide - Smart Notes

Model of the Atom
Historical Models of the Atom
The atomic model has evolved through experimental discoveries. Early models attempted to explain the structure and behavior of atoms based on experimental evidence.
Thomson's Model: Proposed the "plum pudding" model, where electrons are embedded in a positively charged sphere.
Rutherford's Model: Demonstrated that atoms have a small, dense, positively charged nucleus with electrons orbiting around it.
Bohr Model: Suggested that electrons occupy specific orbits (energy levels) around the nucleus and can transition between these levels by absorbing or emitting energy.
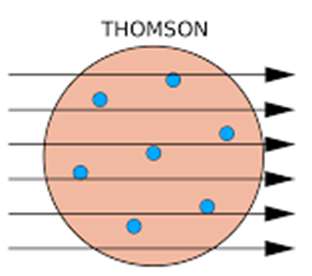
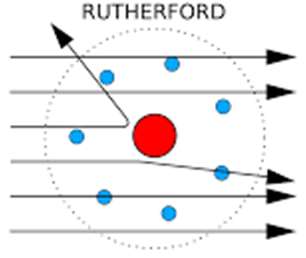
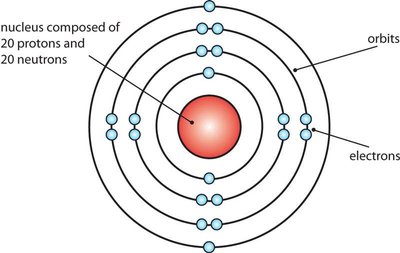
Key Experiments in Atomic Structure
Cathode Ray Tube Experiment: Showed the existence of electrons as negatively charged particles.
Gold Foil Experiment: Revealed the presence of a dense nucleus at the center of the atom.
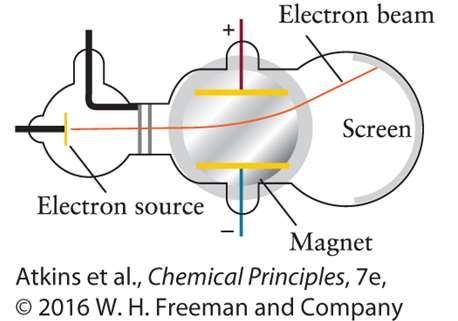
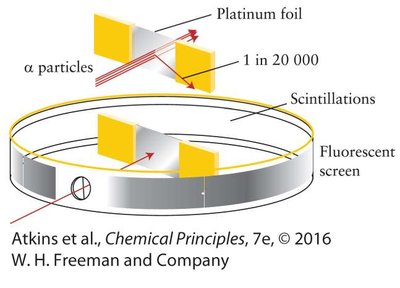
Light and the Electromagnetic Spectrum
Nature of Light
Light is a form of electromagnetic radiation that exhibits both wave-like and particle-like properties. It consists of oscillating electric and magnetic fields and travels through space carrying energy.
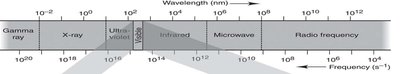
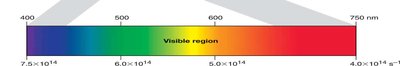
Visible light is only a small part of the electromagnetic spectrum, ranging from about 400 nm (violet) to 750 nm (red).
Other regions include gamma rays, X-rays, ultraviolet, infrared, microwaves, and radio waves.
Wave Properties of Light
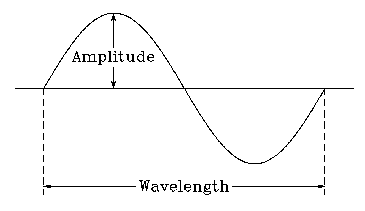
All waves, including light, are characterized by their wavelength, frequency, and amplitude.
Wavelength (\lambda): The distance between two consecutive peaks of a wave (measured in meters or nanometers).
Frequency (\nu): The number of wave cycles passing a point per second (measured in Hz or s-1).
Amplitude: The height of the wave crest, related to the intensity of the radiation.
The relationship between wavelength, frequency, and the speed of light (c) is given by:
where m/s (speed of light in vacuum).
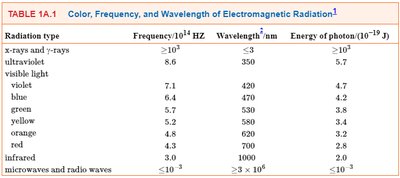
Electromagnetic Spectrum Table
The table below summarizes the frequency, wavelength, and energy of different types of electromagnetic radiation:
Radiation Type | Frequency (1014 Hz) | Wavelength (nm) | Energy of Photon (10-19 J) |
|---|---|---|---|
X-rays and γ-rays | >104 | <3 | >103 |
Ultraviolet | >8.6 | <350 | 5.7 |
Visible light | 7.1–4.3 | 420–700 | 4.7–2.8 |
Infrared | 3.0 | 1000 | 2.0 |
Microwaves and radio waves | <10-3 | >8 × 106 | <10-3 |
Quantization of Energy and the Photoelectric Effect
Blackbody Radiation and Planck's Hypothesis
Classical physics could not explain the emission of light from heated objects (blackbody radiation). Max Planck proposed that energy is quantized and can only be emitted or absorbed in discrete packets called quanta (singular: quantum).
Planck's constant (h): J·s
Energy of a photon:

The Photoelectric Effect
The photoelectric effect is the emission of electrons from a metal surface when light of sufficient frequency shines on it. This phenomenon could not be explained by the wave model of light alone.
Electrons are only ejected if the light has a frequency above a certain threshold frequency (\nu_0).
The kinetic energy of ejected electrons increases with the frequency of the
