 Back
BackAtoms and Elements: Foundations of Modern Atomic Theory
Study Guide - Smart Notes

Atoms and Elements
Introduction to Atoms
Atoms are the fundamental building blocks of matter. The concept of the atom originated from the Greek word atomos, meaning "indivisible." Atoms are the smallest identifiable units of elements and cannot be divided further without losing the properties of the element. There are about 91 naturally occurring elements, with over 20 more created synthetically.
Early Ideas About the Building Blocks of Matter
Early Greek philosophers such as Leucippus and Democritus proposed that matter was composed of small, indestructible particles called atoms. They believed that different types of atoms existed, each with unique shapes and sizes, moving randomly through empty space. However, Plato and Aristotle rejected this idea, suggesting instead that matter was continuous and composed of varying proportions of fire, air, earth, and water.

Later, the scientific method became the standard for investigating the physical world. John Dalton, an English chemist, provided experimental evidence supporting the atomic theory, reviving the ideas of Leucippus and Democritus.

Modern Atomic Theory and Fundamental Laws
The modern atomic theory is based on several key laws derived from experimental observations:
Law of Conservation of Mass: Matter is neither created nor destroyed in a chemical reaction (Antoine Lavoisier).
Law of Definite Proportions: All samples of a given compound have the same proportions of their constituent elements (Joseph Proust).
Law of Multiple Proportions: When two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers (John Dalton).
Dalton’s atomic theory explained these laws by proposing that elements are composed of tiny, indestructible particles (atoms), all atoms of an element are identical, atoms combine in simple ratios to form compounds, and atoms are rearranged (not changed into other elements) during chemical reactions.
Discovery of Subatomic Particles
Although Dalton considered atoms indivisible, later experiments revealed that atoms are composed of smaller particles:
Electrons: Discovered by J.J. Thomson using cathode ray experiments, electrons are negatively charged and much lighter than atoms.
Protons: Positively charged particles found in the nucleus.
Neutrons: Neutral particles in the nucleus, discovered by James Chadwick.
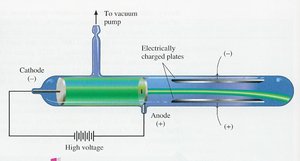
Thomson’s "plum-pudding" model proposed that electrons were embedded in a positively charged sphere. However, Rutherford’s gold foil experiment showed that atoms have a small, dense nucleus containing protons (and later, neutrons), with electrons dispersed in mostly empty space.
Atomic Structure and Isotopes
Atoms are defined by their number of protons (atomic number, Z). The total number of protons and neutrons gives the mass number. Atoms of the same element with different numbers of neutrons are called isotopes. Isotopes have the same atomic number but different mass numbers. The natural abundance of isotopes is relatively constant for each element.
Ions: Losing and Gaining Electrons
Atoms can gain or lose electrons during chemical changes, forming ions:
Cations: Positively charged ions formed by losing electrons (typically metals).
Anions: Negatively charged ions formed by gaining electrons (typically nonmetals).
The Periodic Table and Classification of Elements
The periodic table organizes elements by increasing atomic number and groups elements with similar properties together. Key classifications include:
Metals: Good conductors, malleable, ductile, shiny, tend to lose electrons.
Nonmetals: Poor conductors, not malleable or ductile, tend to gain electrons.
Metalloids: Exhibit mixed properties, often semiconductors.
Special groups include:
Noble gases (Group 8A): Unreactive, stable gases.
Alkali metals (Group 1A): Highly reactive metals.
Alkaline earth metals (Group 2A): Fairly reactive metals.
Halogens (Group 7A): Very reactive nonmetals.
Atomic Mass and Isotopic Abundance
The atomic mass (or atomic weight) of an element is the weighted average of the masses of its naturally occurring isotopes. It is calculated using the formula:
Example: For chlorine, with 75.77% chlorine-35 (34.97 amu) and 24.23% chlorine-37 (36.97 amu):
The Mole Concept and Avogadro’s Number
The mole (mol) is a counting unit for atoms, molecules, or formula units. One mole contains Avogadro’s number () of entities. The molar mass (g/mol) is numerically equal to the atomic or molecular mass in amu.
1 mole of C has a mass of 12 g and contains atoms.
Mole calculations allow conversion between mass, moles, and number of particles.
Sample Mole Calculations
To convert between mass and moles:
To find the number of atoms:
Visualizing Atoms and Molecules
The distinction between atoms and molecules can be illustrated as follows:

Example: A water molecule (H2O) consists of two hydrogen atoms and one oxygen atom bonded together.
