 Back
BackAtoms and Elements: Foundations of Modern Atomic Theory
Study Guide - Smart Notes

Atoms and Elements
Introduction to Atoms
Atoms are the fundamental building blocks of matter. The concept of the atom, derived from the Greek word atomos meaning "indivisible," is central to understanding the composition and behavior of all substances. Atoms are the smallest identifiable units of elements, and all matter is composed of these particles.
Atom: The smallest unit of an element that retains the properties of that element.
Element: A substance composed of only one type of atom; there are about 91 naturally occurring elements and over 20 synthetic ones.
Early Ideas About Matter
Ancient philosophers such as Leucippus and Democritus first proposed that matter was composed of small, indestructible particles called atoms. They believed that different types of atoms existed, each with unique shapes and sizes, moving randomly through empty space. However, Plato and Aristotle rejected this idea, suggesting instead that matter was continuous and composed of varying proportions of fire, air, earth, and water.

Development of Modern Atomic Theory
The scientific approach to understanding matter led to the acceptance of atomic theory. John Dalton provided experimental evidence supporting the existence of atoms and formulated the first modern atomic theory.

Dalton's Atomic Theory:
Each element is composed of tiny, indestructible atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple, whole-number ratios to form compounds.
Atoms are rearranged in chemical reactions but are not changed into other elements.
Key Laws Leading to Atomic Theory
Law of Conservation of Mass
Formulated by Antoine Lavoisier, this law states that matter is neither created nor destroyed in a chemical reaction. The total mass of substances remains constant during chemical changes.
Law of Definite Proportions
Joseph Proust observed that all samples of a given compound have the same proportions of their constituent elements, regardless of source or preparation method. This is also known as the law of constant composition.
Law of Multiple Proportions
John Dalton's law states that when two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers. For example, carbon monoxide (CO) and carbon dioxide (CO2) both contain carbon and oxygen, but in different ratios.
Structure of the Atom
Discovery of Subatomic Particles
Atoms are composed of smaller particles: protons, neutrons, and electrons. J.J. Thomson discovered the electron using cathode ray experiments, showing that electrons are negatively charged and much lighter than atoms.
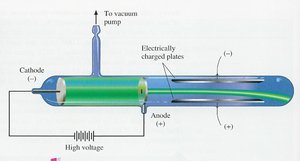
Electron: Negatively charged subatomic particle found in all atoms.
Proton: Positively charged particle found in the nucleus.
Neutron: Neutral particle with a mass similar to the proton, also in the nucleus.
Millikan’s Oil Drop Experiment
Robert Millikan measured the charge of the electron by observing the behavior of oil droplets in an electric field. Combining this with Thomson’s charge-to-mass ratio allowed calculation of the electron’s mass.
Rutherford’s Gold Foil Experiment and the Nuclear Model
Ernest Rutherford’s gold foil experiment demonstrated that atoms have a small, dense, positively charged nucleus surrounded by electrons. Most of the atom’s volume is empty space.
Most of the atom’s mass and all positive charge are in the nucleus.
Electrons occupy the space around the nucleus.
Atoms are electrically neutral overall.
Atomic Number, Mass Number, and Isotopes
Atomic Number (Z)
The atomic number is the number of protons in an atom’s nucleus and defines the element. All atoms of the same element have the same atomic number.
Mass Number (A)
The mass number is the total number of protons and neutrons in an atom.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons and thus different mass numbers. The chemical properties remain similar, but physical properties such as mass differ.
Example: Carbon-12, Carbon-13, and Carbon-14 are isotopes of carbon.
Ions
Formation of Ions
Atoms can gain or lose electrons during chemical changes, forming ions. Cations are positively charged (loss of electrons), and anions are negatively charged (gain of electrons).
The Periodic Table
Organization and Classification
The periodic table arranges elements by increasing atomic number. Elements with similar properties are grouped in columns called groups or families, while rows are called periods.
Metals: Good conductors, malleable, ductile, shiny, tend to lose electrons.
Nonmetals: Poor conductors, not malleable or ductile, tend to gain electrons.
Metalloids: Exhibit properties intermediate between metals and nonmetals.
Special Groups
Noble Gases (Group 8A): Unreactive, stable gases (e.g., helium, neon).
Alkali Metals (Group 1A): Highly reactive metals (e.g., sodium, potassium).
Alkaline Earth Metals (Group 2A): Fairly reactive metals (e.g., calcium, magnesium).
Halogens (Group 7A): Very reactive nonmetals (e.g., fluorine, chlorine).
Atomic Mass and the Mole Concept
Atomic Mass
The atomic mass (or atomic weight) of an element is the weighted average of the masses of its naturally occurring isotopes. It is listed below the element symbol on the periodic table.
Calculation: Multiply the mass of each isotope by its natural abundance (as a decimal), then sum the results.
The Mole and Avogadro’s Number
The mole (mol) is a counting unit for atoms, molecules, or other particles. One mole contains Avogadro’s number () of entities. The molar mass (g/mol) is numerically equal to the atomic or molecular mass in amu.
Example: 1 mole of carbon-12 atoms has a mass of 12 g and contains atoms.
Converting Between Mass, Moles, and Number of Particles
To convert between mass, moles, and number of atoms or molecules, use the molar mass and Avogadro’s number as conversion factors.
Formula:
Formula:
Visualizing Atoms and Molecules
Atoms and molecules can be represented visually to illustrate their composition and structure. For example, a water molecule (H2O) consists of two hydrogen atoms and one oxygen atom bonded together.

