 Back
BackAtoms and Elements: Foundations of Modern Chemistry
Study Guide - Smart Notes

Chapter 2. Atoms and Elements
1. Modern Atomic Theory
Modern atomic theory provides the basis for understanding the composition and behavior of matter. It is founded on several key laws and postulates that describe how atoms combine and interact.
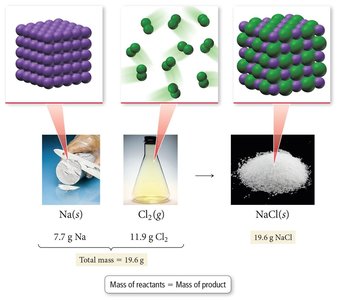
Law of Conservation of Mass: In a chemical reaction, matter is neither created nor destroyed. The total mass of reactants equals the total mass of products. This law supports the idea that matter is composed of small, indestructible particles (atoms).
Law of Definite Proportions: All samples of a given compound have the same proportions of their constituent elements, regardless of source or preparation method.
Law of Multiple Proportions: When two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers.
Dalton's Atomic Theory: Dalton proposed that matter is composed of atoms, each element consists of only one kind of atom, compounds are combinations of atoms in simple ratios, and atoms cannot change into other elements during chemical reactions.
Example: The reaction of sodium and chlorine to form sodium chloride demonstrates the Law of Conservation of Mass.
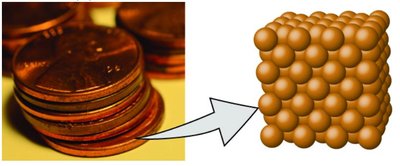
Example: A pre-1982 copper penny contains approximately 3×1022 copper atoms, each with identical chemical properties.
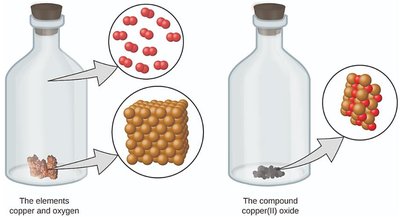
Example: Copper(II) oxide consists of copper and oxygen atoms in a 1:1 ratio, illustrating Dalton's postulate about compounds.

Additional info: Dalton's theory was limited by the lack of knowledge about subatomic particles and isotopes.
2. Atomic Structure
The structure of the atom is fundamental to understanding chemical properties and reactions. Atoms consist of a nucleus and electrons.
Nucleus: The core of the atom, containing protons (positively charged) and neutrons (neutral), and accounting for most of the atom's mass.
Electrons: Negatively charged particles with very little mass, found in regions around the nucleus.
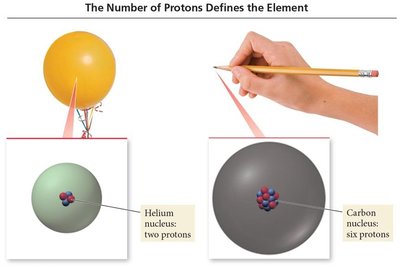
Atomic Number (Z): The number of protons in the nucleus, which defines the element.
Isotopes: Atoms of the same element with different numbers of neutrons, resulting in different mass numbers (A).
Ions: Atoms that have gained or lost electrons, resulting in a net charge. Cations are positively charged, anions are negatively charged.
Example: The number of protons distinguishes helium (2 protons) from carbon (6 protons).
Nuclide Symbol: Isotopes are represented as , where X is the chemical symbol, A is the mass number, and Z is the atomic number.
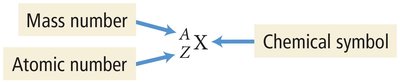
Alternative Notation: Isotopes can also be written as X-A (e.g., Ne-20).
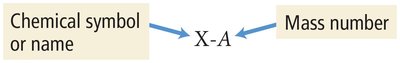
Example: Neon has three naturally occurring isotopes, each with different numbers of neutrons and natural abundances.
Symbol | Number of Protons | Number of Neutrons | A (Mass Number) | Natural Abundance (%) |
|---|---|---|---|---|
Ne-20 or | 10 | 10 | 20 | 90.48 |
Ne-21 or | 10 | 11 | 21 | 0.27 |
Ne-22 or | 10 | 12 | 22 | 9.25 |
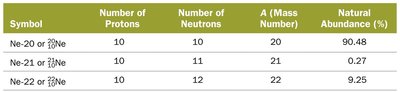
Additional info: Isotopes of an element generally exhibit the same chemical behavior.
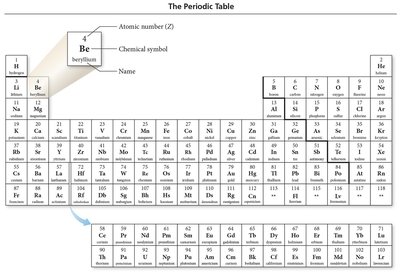
3. The Periodic Table
The periodic table organizes elements by increasing atomic number and groups them based on similar chemical properties. It is a powerful tool for predicting element behavior and ion formation.
Groups (Columns): Elements in the same group have similar properties.
Periods (Rows): Elements in the same period have the same number of electron shells.
Classification: Elements are classified as metals, nonmetals, or metalloids.
Metals: Good conductors, malleable, ductile, tend to lose electrons.
Nonmetals: Poor conductors, not malleable or ductile, tend to gain electrons.
Metalloids: Exhibit mixed properties, often semiconductors.
Example: The periodic table can be used to predict the charge of ions formed by main-group elements.

Additional info: Alkali metals (group 1A) form 1+ ions, alkaline earth metals (group 2A) form 2+ ions, halogens (group 7A) form 1– ions, and chalcogens (group 6A) form 2– ions.
4. Atomic Mass
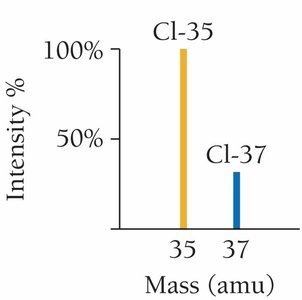
Atomic mass is the average mass of an element's isotopes, weighted by their natural abundance. It is measured in atomic mass units (amu) and is determined using mass spectrometry.
Mass Spectrometer: An instrument that separates ions by mass and measures their abundance.
Mass Spectrum: A plot of intensity (%) versus mass/charge ratio, showing the relative abundance of isotopes.
Calculation: Atomic mass is calculated as the sum of the products of each isotope's mass and its fractional abundance.

Formula:
Example: Chlorine's atomic mass is calculated from its two isotopes:
Cl-35: 75.77% abundance, mass 34.97 amu
Cl-37: 24.23% abundance, mass 36.97 amu
Weighted average: amu
5. Molar Mass: Counting Atoms by Weighing Them
The mole is a fundamental unit for counting atoms, molecules, or ions. Molar mass allows conversion between mass and number of particles.
Mole: 1 mole = particles (Avogadro's number).
Molar Mass: The mass of 1 mole of an element (in grams) is numerically equal to its atomic mass (in amu).
Conversions: Use molar mass and Avogadro's number to convert between mass, moles, and number of atoms.

Example Calculations:
Number of atoms in 3.25 mol Fe: atoms
Moles of Ag in 7.811 g: mol
Number of Pb atoms in 0.224 g: atoms
Additional info: The lighter the atom, the less mass in 1 mole of atoms.
